|
Discussion Board
Paneles de Discussión
Paneais de Discussio
Free Papers
Comunicaciones libres
Comunicaçoes livres
Home cin2003
Volver al Inicio cin2003
Voltar ao inicio cin2003
|
CEREBRAL WHITE MATTER LESIONS IN ESSENTIAL HYPERTENSION
Cristina Sierra, MD, PhD
Hypertension Unit. Hospital Clinic.
Institut d´Investigacions Biomédiques August Pi i Sunyer (IDIBAPS).
University of Barcelona. Spain
INTRODUCTION
Since the introduction of highly sensitive brain MRI more than 15 years ago, the presence of cerebral hyperintensities in the deep and subcortical white matter has been a common finding in elderly people [1]. The pathogenesis of cerebral white matter lesions (WML) is poorly understood. However, most studies have shown that age, hypertension, diabetes mellitus and a history of stroke or heart disease are the most important factors related to the presence of WML [1]. Indeed, besides age, hypertension is constantly reported to be the main risk factor for cerebral WML. The association between WML and hypertension may be mediated through several different pathogenetic pathways, and in this sense, the presence of WML has been reported to be associated with the severity of high blood pressure (BP) values [2], lack of blood pressure control in treated hypertensive patients [3], and nocturnal BP dip [4]. In addittion, the relationship between some genetic polymorphisms and the presence of WML in hypertensive patients has been recently reported [5]. The presence of cerebral WML is an important prognostic factor for the development of stroke [6] and also for cognitive impairment [7], and could be considered an early marker of brain damage in essential hypertensive patients (Figure 1).
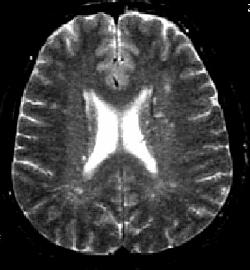 Figure 1. Brain-Magnetic Resonance Imaging (axial plane T2)
Figure 1. Brain-Magnetic Resonance Imaging (axial plane T2)
showing both white matter hyperintensities directly adjacent to the ventricles (periventricular lesions),
and confluent white matter hyperintensities at some distance from the ventricles (focal lesions).PATHOGENESIS OF CEREBRAL WML
The main hypothesis regarding the association between high BP and ischaemic WML is that long-standing hypertension causes lipohyalinosis of the media and thickening of the vessel walls, with narrowing of the lumen of the small perforating arteries and arterioles which nourish the deep white matter [1]. The perforating vessels, which originate from cortical and leptomeningeal arteries, have a relatively poor anastomotic system, which makes the white matter vulnerable to cerebral ischemia. In this sense, low BP has also been reported to be a risk factor for WML [1]. Hypertension may also cause disturbances in the blood-brain barrier, which may cause lesions in the white matter by cerebral edema, by activation of astrocytes, or by destructive enzymes or other poisons which pass through the damaged vessel walls [1].
Postmortem studies have indicated that WML seen on MRI scans are associated with degenerative changes in arterioles that are related to atherosclerosis, suggesting that cerebral arteriosclerosis of the penetrating vessels is the main factor in the pathogenesis of ischemic WML [1]. Bots et al [8] reported that WML were related to atherosclerosis, indicated by increased common carotid intima-media thickness and carotid plaques. In the same way, de Leeuw et al [9] showed that the presence of aortic atherosclerosis during midlife, assessed on abdominal radiographs, was significantly associated with the presence of periventricular WML 20 years later.
GENETIC FACTORS AND CEREBRAL WML
Genetic risk factors have been implicated in the presence and severity of WML but remain undetermined so far. A recent genetic study on elderly twins indicated that the susceptibility to white matter hyperintensity on brain MRI was largely determined by genetic factors [10]. Genes contributing to interindividual variation in BP levels and essential hypertension may play a role in the etiology of WML or stroke, either through their effects on BP levels or through separate pathways. In this sense, the renin-angiotensin system is an example of a system that may be involved in the pathogenesis of both hypertension and arteriosclerosis. Kario et al [11] found a positive association between angiotensin converting enzyme D allele and the presence of both silent and clinically overt stroke in Japanese hypertensives. Sierra et al [5] reported an association between the presence of the DD genotype and/or the D allele of the angiotensin converting enzyme gene and WML in asymptomatic middle-aged hypertensive patients.
CEREBRAL BLOOD FLOW AND WML
High BP influences the cerebral circulation causing adaptative vascular changes. Thus, hypertension influences the autoregulation of cerebral blood flow by shifting both lower and upper limits of autoregulatory capacity towards a higher blood pressure, and hypertensive patients may be especially vulnerable to episodes of hypotension [1], which may play a role in the development of WML. However, most studies have found no significant changes in resting cerebral blood flow in both normotensive and hypertensive individuals with silent WML. On the contrary, with relation to vasomotor reactivity (or vasodilatory capacity) and WML there are controversial results. Kuwabara et al [12] reported a close relationship between cerebral hemodynamic reserve capacity, measured by positron emission tomography, and the severity of WML in hypertensive patients. Bakker et al [13] confirmed the association between decreased vasomotor reactivity and WML, measured by means of a transcranial doppler in 73 elderly individuals, 56% of whom were hypertensives. Conversely, Chamorro et al [14] showed a preserved vasomotor reactivity in 41 patients (71% hypertensives) with silent WML and first-ever lacunar infarction, although they had an increased cerebrovascular tone, measured by transcranial doppler.
PREVALENCE OF CEREBRAL WML
Several studies have examined the prevalence of WML in both normotensive and hypertensive subjects. The ARIC study [3] reported a 24.6% prevalence of WML among individuals aged 55-72 years, 49% of whom were hypertensives. The Cardiovascular Health Study [15] found a prevalence of 33.3% in individuals aged 65 years or older, 44% of whom were hypertensives. The prevalence was 27% in the Rotterdam Study [16], which included individuals aged 65-84 years, 39% of whom were hypertensives. Shimada et al [2] studied 28 normotensives and 20 hypertensives aged 59-83 years and found a prevalence of advanced WML of 25% and 40%, respectively. The study of Goldstein et al [17], performed in 144 normotensive individuals aged 55 to 79 years (10% with casual elevations of BP), showed a 54.9% prevalence of WML. Differences in the prevalence of WML among studies may be due to subtle variations in WML assessment, but especially to the different impact of risk factors, such as age and hypertension, which are influenced by the subject criteria selection. In this sense, most studies included both normotensive and hypertensive patients (untreated and treated), or subjects with a wide range of ages or only elderly people.
CEREBRAL WML, BLOOD PRESSURE, AND BLOOD PRESSURE VARIABILITY
The association between hypertension and WML has been established in cross-sectional [1-3,15,16] and longitudinal studies [18]. However, some reports have suggested that this relationship is only evident when 24-hour ABPM is used for the assessment of BP. In this sense, Goldstein et al [17] found a correlation between WML and office systolic, but not diastolic BP, in a group of elderly normotensive subjects. Conversely, the severity of WML correlated with both systolic and diastolic BP, measured by ABPM. In a group of mixed normotensives, "white coat" hypertensives and sustained hypertensives, Shimada et al [2] also found a correlation between the number of lacunae and periventricular hyperintensities with 24-hour BP, but not with office BP.
Concerning circadian pattern of BP, Shimada et al [4] reported that both nondippers and extreme dippers had significantly more silent cerebrovascular damage (measuring both lacunae and WML) than dippers. Although BP variability has been related to target organ damage in hypertension, its relationship with cerebral alterations has not been established. A previous report by Goldstein et al [17] suggested a higher standard deviation of awake SBP in patients with more severe WML. On the other, neither the circadian rhythm nor the long-term variability of BP were related to WML in a group of 66 middle-aged never treated hypertensive patients [19].
COGNITIVE FUNCTION AND CEREBRAL WML
Several studies have shown a correlation between BP and cognitive function in elderly people [20]. In adition, various studies have shown an association between the presence of WML and cognitive function in both normotensive and hypertensive elderly populations [15,21]. It is unclear whether the impact of elevated BP on cognitive decline in late-life is mediated through its chronic and negative effect on the structural characteristics of the brain. Skoog et al [22] reported an association between elevated blood pressure at age 70 and the development of dementia 10-15 years later, while patients with WML at age 85 had a higher BP at age 70, suggesting that previously increased BP may increase the risk of dementia by inducing small-vessel disease and WML. In the same way, Swan et al [23] showed that midlife SBP is a significant predictor of WML and decline in cognitive function.
CONCLUSION
Several pathologic studies have suggested that cerebral WML are caused by arteriolosclerosis, for which hypertension, besides aging, is the main risk factor. Indeed, hypertensive patients have higher rate and extent of areas of WML compared with controls, and also with treated and controlled hypertensives. In addition, some studies have recently reported an association between genetic factors and WML. On the other hand, it is known that hypertension is the main risk factor for stroke and vascular dementia. Similarly, the presence of cerebral WML is an important prognostic factor for the development of stroke and also for cognitive impairment. Therefore, the presence of cerebral WML could be considered an early marker of brain damage in hypertensive patients.
Prospective studies with larger sample size are needed to better assess the relationship between high BP and the presence of WML and their possible complications, as well as the role of different antihypertensive drugs in controlling WML presence and progression.
REFERENCES
- Pantoni L, Garcia JH. The significance of cerebral white matter abnormalities 100 years after Binswanger´s report. Stroke 1995;26:1293-1301.
- Shimada K, Kawamoto A, Matsubayashi K, Ozawa T. Silent cerebrovascular disease in the elderly. Correlation with ambulatory pressure. Hypertension 1990;16:692-699.
- Liao D, Cooper L, Cai J, Toole JF, Bryan N, Hutchinson RG, Tyroler HA. Presence and severity of cerebral white matter lesions and hypertension, its treatment, and its control. The ARIC Study. Stroke 1996;27:2262-2270.
- Kario K, Matsuo T, Kobayashi H, Imiya M, Matsuo M, Shimada K. Nocturnal fall of blood pressure and silent cerebrovascular damage in elderly hypertensive patients. Advanced silent cerebrovascular damage in extreme dippers. Hypertension 1996;27:130-135.
- Sierra C, Coca A, Gómez-Angelats E, Poch E, Sobrino J, de la Sierra A. Renin-angiotensin system genetic polymorphisms and cerebral white matter lesions in essential hypertension. Hypertension 2002;39:343-347.
- van Swieten JC, Kapelle LJ, Algra A, van Latum JC, Koudstaal PJ, van Gijn J. Hypodensity of the cerebral white matter in patients with transient ischemic attack or minor stroke: influence on the rate of subsequent stroke. Dutch TIA Trial Study Group. Ann Neurol 1992;32:177-183.
- De Groot JC, de Leeuw FE, Oudkerk M, van Gijn J, Hofman A, Jolles J, Breteler MMB. Periventricular cerebral white matter lesions predict rate of cognitive decline. Ann Neurol 2002;52:335-341.
- Bots ML, van Swieten JC, Breteler MMB, de Jong PTVM, van Gijn J, Hofman A, Grobbee DE. Cerebral white matter lesions and atherosclerosis in the Rotterdam Study. Lancet 1993;341:1232-1237.
- de Leeuw FE, de Groot JC, Oudkerk M, Witteman JCM, Hofman A, van Gijn J, Breteler MMB. Aortic atherosclerosis at middle age predicts cerebral white matter lesions in the elderly. Stroke 2000;31:425-429.
- Carmelli D, DeCarli C, Swan GE, Jack LM, Reed T, Wolf PA, Miller BL. Evidence for genetic variance in white matter hyperintensity volume in normal elderly male twins. Stroke 1998;29:1177-1181.
- Kario K, Kanai N, Saito K, Nago N, Matsuo T, Shimada K. Ischemic stroke and the gene for angiotensin-converting enzyme in japanese hypertensives. Circulation 1996;93:1630-1633.
- Kuwabara Y, Ichiya Y, Sasaki M, Yoshida T, Fukumura T, Masuda K, Ibayashi S, Fujishima M. Cerebral blood flow and vascular response to hypercapnia in hypertensive patients with leukoaraiosis. Ann Nucl Med 1996;10;293-298.
- Bakker SLM, de Leeuw FE, de Groot JC, Hofman A, Koudstaal PJ, Breteler MMB. Cerebral vasomotor rectivity and cerebral white matter lesions in the elderly. Neurology 1999;52:578-583.
- Chamorro A, Pujol J, Saiz A, Vila N, Vilanova JC, Alday M, Blanc R. Periventricular white matter lucencies in patients with lacunar stroke. Arch Neurol 1997;54:1284-1288.
- Longstreth WT, Manolio TA, Arnold A, Burke GL, Bryan N, Jungreis ChA, Enright PL, O´Leary D, Fried L, for the Cardiovascular Health Study Collaborative Research Group. Clinical correlates of white matter findings on cranial magnetic resonance imaging of 3301 elderly people. The Cardiovascular Health Study. Stroke 1996;27:1274-1282.
- Breteler MMB, van Swieten JC, Bots ML, Grobbee DE, Claus JJ, van den Hout JHW, van Harskamp F, Tanghe HLJ, de Jong PTVM, van Gijn J, Hofman A.. Cerebral white matter lesions, vascular risk factors, and cognitive function in a population-based study: The Rotterdam Study. Neurology 1994;44:1246-1252.
- Goldstein IB, Bartzokis G, Hance DB, Shapiro D. Relationship between blood pressure and subcortical lesions in healthy elderly people. Stroke 1998;29:765-772.
- Dufouil C, de Kersaint-Gilly A, Besancon V, Levy C, Auffray E, Brunnereau L, Alperovitch A, Tzourio C. Longitudinal study on blood pressure and white matter hyperintensities. The EVA MRI cohort. Neurology 2001;56:921-926.
- Sierra C, de la Sierra A, Mercader J, Gómez-Angelats E, Urbano-Márquez A, Coca A. Silent cerebral white matter lesions in middle-aged essential hypertensive patients. J Hypertens 2002;20:519-524.
- Launer LJ, Masaki K, Petrovitch H, Foley D, Havlik RJ. The association between midlife blood pressure levels and late-life cognitive function. JAMA 1995;274:1846–1851.
- De Groot JC, de Leeuw FE, Oudkerk M, van Gijn J, Hofman A, Jolles J, Breteler MMB. Periventricular cerebral white matter lesions predict rate of cognitive decline. Ann Neurol 2002;52:335-341.
- Skoog I, Lernfelt B, Landahl S, Palmertz B, Andreasson LA, Nilsson L, Persson G, Oden A, Svanborg A. 15-year longitudinal study of blood pressure and dementia. Lancet 1996;347:1141-1145.
- Swan GE, DeCarli C, Miller BL, Reed T, Wolf PA, Jack LM, Carmelli D. Association of midlife blood pressure to late-life cognitive decline and brain morphology. Neurology 1998;51:986-993.
|
