
Temperature and Hemodialysis
Daniel Schneditz, Ph.D.
Department of Physiology, University of Graz
Graz, Austria
E-mail: daniel.schneditz@uni-graz.at
| DISCUSSION BOARD |
Introduction
Temperature is such a natural variable in human biology and physiology that at first sight it appears trivial and obvious to discuss its effects, especially in hemodialysis. However, even though the impact of cool dialysate with temperatures around 35 °C on cardiovascular stability has been known since the first studies by Maggiore et al. [1] it is only now that the relationship to other aspects of hemodialysis and to the different blood purification modes is being better understood [2].
Why is temperature important in hemodialysis?
Dialysate temperature
Except for machines which operate with a batch of pre-warmed dialysate, all hemodialysis machines are equipped with a system to control for a desired dialysate temperature. Until recently a dialysate temperature of 37°C was considered adequate for everyday hemodialysis. Even if a dialysate temperature of 37°C is somewhat higher than the average physiologic core temperature, the excess was accepted as a rough compensation for unavoidable heat losses in the venous part of the extracorporeal circulation.
Since the first reports in the early eighties by Maggiore et al. [1] many studies have confirmed that a low dialysate temperature in the range of 34 to 35.5°C improves intradialytic hemodynamic stability when compared to dialysate temperature set at 37°C or higher [3]. Lower dialysate temperature improves cardiac contractility [4] and increases venous tone. For example, in a prospective study done in 11 patients cool dialysis at 35.5°C significantly increased the lowest intra-dialytic as well as the post-dialytic blood pressure and significantly reduced the number of nursing interventions as well as the volume of saline infused for intradialytic hypotension [5]. Fine et al. also observed that especially patients with low body temperatures had the highest benefit from cool dialysate [6]. So why does one dialysate temperature not fit all?
Extracorporeal heat flow
Dialysate temperature (Tdia) is an important factor in thermal balance during hemodialysis. The direct thermal effects of hemodialysis depend on the amount of heat removed (negative sign) or delivered (positive sign) to the patient during a treatment. Extracorporeal heat flow (JV, in W) is given by the arterio-venous temperature gradient in the extracorporeal circulation (Tart-Tven, in °C), extracorporeal blood flow (Qb, in mL/s) corrected for ultrafiltration rate (UFR, in mL/s), and the product of the material constants cr (3.81, in J °C-1 cm-3) [7] [8] [9]:
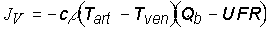 | Eq. 1 |
The temperature of venous blood returning to the patient (Tven) is controlled by dialysate temperature (Tdia) but it is also determined by the passage of blood through the venous line which is exposed to the cool environment. Blood residence time increases as blood flow (Qb) decreases so that at low blood flows Tven may fall by more than 1 °C below Tdia. with low blood flows of 150-250 mL/min [10]. However, the amount of cooling (Jven, in W) provided by the venous blood line at different blood flows is almost independent of Qb and can be viewed as constant (const.) so that Eq. 1 can be written in terms of arterial and dialysate temperatures and a constant term of approximately 7 to 15 W, depending on the insulation characteristics of the venous blood line [11]:
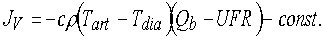 | Eq. 2 |
The temperature of arterial blood (Tart) is determined by body temperature.
Body temperature
In healthy subjects the temperature of the deep tissues of the body, the so-called core, remains almost exactly constant within ±0.5ºC, day in and day out with a normal range for rectal measurements from 36.2 to 37.7ºC [12]. However, there is a large number of dialysis patients with a body temperature at or below the lower limit of normal. Pre-dialysis temperatures measured by infrared tympanic thermometry were less than 36ºC in 28 out of 128 dialysis patients [6]. Mean pre-dialysis rectal temperature was 36.2±0.5ºC in 13 patients so that about half of the patients must have had core temperatures less than 36.2ºC [13]. In a study utilizing the blood temperature monitor (BTM, Fresenius Medical Care Deutschland G.m.b.H., Bad Homburg, Germany), body temperature varied from 36.1 to 37.4ºC which almost covers the normal range [14]. Nine out of 25 patients had a body temperature of less than 36.5ºC. In a different study done in 27 patients utilizing the same temperature monitor, arterial line temperature was 36.2±0.3ºC (range 35.6 to 36.7ºC) [9].
The explanation for the lower body temperature in some patients remains speculative. It has been hypothesized that the degree of uremia may be related to hypothermia but there was no effect of duration of hemodialysis, incidence of diabetes, concentration of hemoglobin, serum creatinine, urea or albumin [6]. However, correlation of body temperature with the dose of delivered dialysis has not been published.
Since body temperature may vary by as much as 1° C in different patients, it is evident from Eqs. 1 and 2 that two dialyses done at the same blood flow such as 400 mL/min and at the same dialysate temperature may lead to a difference in JV of up to 25W. This is almost one third of the patient's energy expenditure and it is likely to have a major impact on physiologic temperature control.
Energy expenditure
It is instructive to compare the magnitude of thermal energy flows in the extracorporeal system to the energy expenditure of the patient. Resting energy expenditure (REE, in W) in healthy adults can be estimated from anthropometric data using the Harris-Benedict equation [15]:
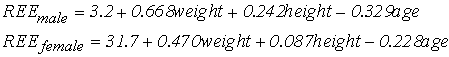 | Eq. 3 |
where weight, height, and age are given in kg, cm, and years, respectively. REE for the average adult is in the range of 75 W. Since both kidneys cover approximately 10% of REE [16] the question arises whether the anthropometric formula is applicable to chronic renal failure patients. However, specific REE (in W/kg) measured in 52 hemodialysis patients (1.14±0.06 W/kg) was not different from that of healthy subjects (1.30±0.15 W/kg) [17]. It appears as if the expected reduction in REE by the loss in kidney function in hemodialysis patients was offset by a higher metabolic rate of other body tissues. It can be concluded that the Harris-Benedict equation remains applicable for the average hemodialysis patient.
The measurement of extracorporeal heat flow has been helpful in clarifying some myths about the hemodynamic benefits of convective vs. diffusive treatments. For a long time convective treatments such as hemofiltration have been known to provide better hemodynamic stability than diffusive treatments, even when controlled for all aspects of volume and solute balance as well as for the type of membranes used. However, under-recognized at that time, convective treatment modes markedly differ in extracorporeal heat balance. Approximately one third of thermal energy expenditure is lost during isolated ultrafiltration because blood passing the extracorporeal circulation is not exposed to warm dialysate [18]. But when convective and diffusive treatment modes were matched for changes in body temperatures and/or extracorporeal heat flow, differences in vascular reactivity such as forearm vascular resistance, forearm venous tone, and arterial blood pressure between treatment modes disappeared [19], [18]. In a study with 10 dialysis patients Keijman et al. had to decrease dialysate temperature to 34.8 °C to match the extracorporeal heat flow of -31 W obtained by isolated ultrafiltration [20]. Thus, convective treatment modes provide a significant amount of extracorporeal cooling which prevents a rise in body temperature.
Heat accumulation during hemodialysis
Contrary to uremic solutes, which accumulate between treatments, there is a potential for thermal energy to accumulate within a hemodialysis treatment. Essentially, there are three possibilities for intradialytic heat accumulation:
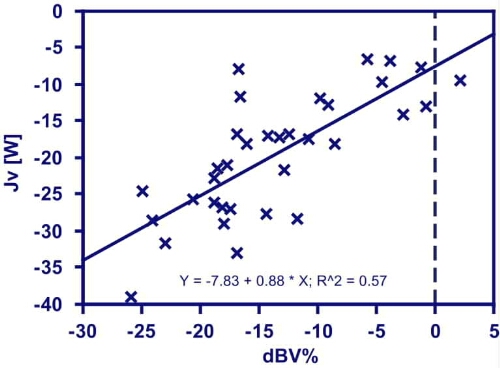
The hemodynamic hypothesis of intradialytic heat accumulation is supported by recent observations by Rosales et al. showing that the amount of thermal energy to be removed for an isothermic hemodialysis correlates with ultrafiltration requirements and with relative blood volume changes, two measures of hemodynamic perturbation [9], [23] (Fig. 1). In one of these studies done in 27 patients extracorporeal thermal flow rate was 17±6 W, i.e. 28±10% of estimated energy expenditure as determined from an anthropometric relationship (Eq. 3).
Feedback control of patient temperature
The rationale of temperature control is to prevent heat accumulation which increases body temperature in the patient during hemodialysis. It follows from the discussion above that dialysate temperature should be individualized and chosen with regard to actual patient temperature, blood flow and treatment mode such as hemodialysis and hemodiafiltration. Above that, it is not sufficient to maintain a constant dialysate temperature throughout the treatment but to adjust dialysate temperature to control for a defined patient temperature.
BTM control
The blood temperature monitor (BTM) is a module built to fit current Fresenius hemodialysis machines (Fig. 2). Short sections of the extracorporeal circulation are inserted into arterial and venous measuring heads equipped with sensors to measure arterial and venous blood temperatures as described elsewhere. The arterial and venous tube sections, which have to meet certain specifications to fit into the measuring heads, are located within a given distance from the access. The blood temperature at the access is then calculated for given insulation and environmental conditions using the extracorporeal blood flow measured by the dialysis machine. The precision of the temperature measurement is better than 0.1°C for blood flows above 120 mL/min. Tart and Tven are measured with a sampling period of 15 s. Recirculation is measured in 30 minute intervals utilizing the automatic thermodilution option of the device which is described elsewhere [24].

Patient temperature was one of the first physiologic variables to be automatically controlled during hemodialysis by the T-control mode offered by the BTM. The T-control mode requires the prescription of an hourly change in body temperature. To control for a constant body temperature throughout a treatment one would have to prescribe a temperature change rate of ±0.00°C/h. The BTM controller uses the error signal between desired and actual change in temperature to actuate a bounded change in dialysate temperature (Tdia) which changes the temperature of the venous (Tven) blood returning to the patient thereby changing the extracorporeal heat flow (Fig. 3). The BTM can also be operated in an E-control mode, which controls for the rate of thermal energy removal (dE/dt, in kJ/h). Even if this type of control affects patient temperature it actually controls thermal flow rate, which is not a physiologic variable. Thus, it is not typical for physiologic feedback control.
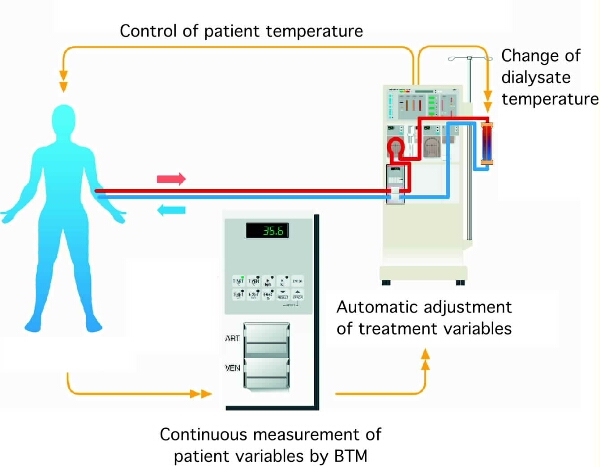
Fig. 3. Temperature control by the BTM. Measurement of arterial and venous line temperatures by the BTM in a setup to control patient temperature by automatic adjustment of dialysate temperature (reproduced with permission by Fresenius Medical Care Deutschland GmbH).
Optimum profile for body temperature
It follows from the analysis of thermal balance in convective treatment modes that hemodynamic stability is improved when body temperature is prevented from rising during treatments and when a large fraction of thermal energy produced in the patient is removed from the body through the extracorporeal circulation. When concerned about hemodynamic stability it is probably best to prevent an increase in body temperature. Since a decrease in body temperature is likely to elicit powerful compensatory mechanisms such as an increase in metabolic rate, maintaining body temperature at a constant level is probably a good choice.
Summary
One dialysate temperature does not fit all patients mainly because of the wide range of core temperatures observed in hemodialysis patients and because small arterio-venous temperature gradients operating over prolonged periods of time exert significant effects on thermal balance. Even when the treatment is carried out without heat exchange in the extracorporeal circulation, thermal energy tends to accumulate in hemodialysis patients most likely because of reduced heat dissipation from the body shell to the environment. The reduced heat dissipation can be linked to hemostatic control mechanisms triggered by ultrafiltration induced hypovolemia. Up to 50% of energy expenditure needs to be removed from the patient during ultrafiltration to maintain a constant body temperature. Physiologic thermoregulation which interferes with hemostatic control is predominantly determined by core body temperature so that blood purification techniques which have a considerable impact on thermal balance should aim at a constant body temperature rather than at a constant dialysate temperature. Control of body temperature is easily achieved by the BTM which automatically adjust for variable blood flows, temperatures and treatment modes.
Bibliography
1. Maggiore Q, Pizzarelli F, Zoccali C, Sisca S, Nicolo F, Parlongo S: Effect of extracorporeal blood cooling on dialytic arterial hypotension. Proc Eur Dial Transplant Assoc 18:597-602, 1981
2. Sherman RA: Advancing the cold front. Am J Kidney Dis 36:412-414, 2000
3. Sherman RA, Rubin MP, Cody RP, Eisinger RP: Amelioration of hemodialysis-associated hypotension by the use of cool dialysate. Am J Kidney Dis 5:124-127, 1985
4. Levy FL, Grayburn PA, Foulks CJ, Brickner ME, Henrich WL: Improved left ventricular contractility with cool temperature hemodialysis. Kidney Int 41:961-965, 1992
5. Cruz DN, Mahnensmith RL, Brickel HM, Perazella MA: Midodrine and cool dialysate are effective therapies for symptomatic intradialytic hypotension. Am J Kidney Dis 33:920-926, 1999
6. Fine A, Penner B: The protective effect of cool dialysate is dependent on patients' predialysis temperature. Am J Kidney Dis 28:262-265, 1996
7. Krämer, M. and Polaschegg, H. D. Control of blood temperature and thermal energy balance during hemodialysis. Proc IEEE EMBS. 14, 2299-2300, 1992
8. Schneditz D, Martin K, Krämer M, Kenner T, Skrabal F: Effect of controlled extracorporeal blood cooling on ultrafiltration induced blood volume changes during hemodialysis. J Am Soc Nephrol 8:956-964, 1997
9. Rosales LM, Schneditz D, Morris AT, Rahmati S, Levin NW: Isothermic hemodialysis and ultrafiltration. Am J Kidney Dis 36:353-361, 2000
10. Morris AT, Schneditz D, Fan Z, Kaufman AM, Levin NW: Dialysate temperature is not the sole determinant of extracorporeal blood cooling during hemodialysis (HD). (abstract) J Am Soc Nephrol 7:1414, 1996
11. Schneditz D: Temperature and thermal balance in hemodialysis. Sem Dial 14:357-364, 2001
12. Altman PL, Dittmer SD: Metabolism, Bethesda, MD, FASEB, 1968
13. Lange H, Krautwald E, Krautwald G, Ebel H: The effect of extracorporeal haemodialysis on energy turnover. Proc EDTA-ERCA 22:106-110, 1985
14. Krämer, M. and Polaschegg, H. D. The relevance of thermal effects during hemodialysis. Friedman, E. A. and Beyer, M. M. American Society for Artificial Internal Organs, 39th Annual Meeting. M84. Philadelphia, PA, J.B. Lippincott, 1993
15. Harris A, Benedict FG: Publication 279; A biometric study of human basal metabolism in man, Washington, DC, Carnegie Institute, 1919
16. Gallagher D, Belmonte D, Deurenberg P, Wang Z, Krasnow N, Pi-Sunyer FX, Heymsfield SB: Organ-tissue mass measurement allows modeling of REE and metabolically active tissue mass. Am J Physiol 275:E249-E258, 1998
17. Kuhlmann U, Schwickardi M, Lange H: Energy expenditure in patients with renal failure. (abstract) Nephrol Dial Transplant 15:A50, 2000
18. van der Sande FM, Gladziwa U, Kooman JP, Bocker G, Leunissen KM: Energy transfer is the single most important factor for the difference in vascular response between isolated ultrafiltration and hemodialysis. J Am Soc Nephrol 11:1512-1517, 2000
19. van Kuijk WH, Hillion D, Savoiu C, Leunissen KM: Critical role of the extracorporeal blood temperature in the hemodynamic response during hemofiltration. J Am Soc Nephrol 8:949-955, 1997
20. Keijman JM, van der Sande FM, Kooman JP, Leunissen KM: Thermal energy balance and body temperature: comparison between isolated ultrafiltration and haemodialysis at different dialysate temperatures. Nephrol Dial Transplant 14:2196-2200, 1999
21. Gotch FA, Keen ML, Yarian SR: An analysis of thermal regulation in hemodialysis with one and three compartment models. ASAIO Transactions 35:622-624, 1989
22. Amberson WR: Physiologic adjustments to the standing posture. Univ Md Sch Med Bull 27:127-145, 1943
23. Rosales L, Schneditz D, Ronco C, Levin NW: Heat accumulation during hemodialysis (HD) depends on blood volume reduction. (abstract) J Am Soc Nephrol 11:296A, 2000
24. Kaufman AM, Krämer M, Godmere RO, Morris AT, Amerling R, Polaschegg HD, Levin NW: Hemodialysis access recirculation (R) measurement by blood temperature monitoring (BTM) - A new technique. (abstract) J Am Soc Nephrol 2:324, 1991