|
Discussion Board
Paneles de Discussión
Paneais de Discussio
Free Papers
Comunicaciones libres
Comunicaçoes livres
Home cin2003
Volver al Inicio cin2003
Voltar ao inicio cin2003
|
CALCITRIOL AS A NEGATIVE REGULATOR OF THE RENIN-ANGIOTENSIN SYSTEM
Yan Chun Li
Department of Medicine, University of Chicago.
Chicago, USA.
cyan@medicine.bsd.uchicago.edu
Introduction
The renin-angiotensin system (RAS) plays a crucial role in the regulation of electrolyte, extracellular volume and blood pressure homeostasis. The primary physiological role of the vitamin D endocrine system is to regulate calcium homeostasis. As the most active metabolite of vitamin D, calcitriol [1,25-dihydroxyvitamin D3, 1,25(OH)2D3] is responsible for most of the biological actions of the vitamin D endocrine system, which are mediated by the vitamin D receptor (VDR), a member of the nuclear receptor superfamily 1. Studies using genetically mutant mice have demonstrated that the vitamin D endocrine system has additional physiological functions beyond calcium homeostasis, which include immune responses, reproduction, mammary gland development and hair growth 2-6. Our recent works show that calcitriol also functions as a negative endocrine regulator of the RAS and thus plays an important role in the regulation of the renocardiovascular functions 7, 8.
Clinical and epidemiological evidence linking vitamin D-deficiency with hypertension
Accumulating evidence from epidemiological and clinical studies in the last decades has suggested a connection between vitamin D and blood pressure. As ultraviolet (UV) irradiation is essential for the cutaneous production of vitamin D, circulating vitamin D levels are greatly influenced by geographic locations, seasonal changes and skin pigmentations. Data obtained from the INTERSALT study centers reveal a linear correlation between the rise in blood pressure or the prevalence of hypertension and the latitudes north or south of the equator 9. Data from a national survey in China also demonstrate that the prevalence of hypertension and stroke incidence display a high-to-low gradient from the north to the south of the country 10. Seasonal variations in blood pressure have been reported in temperate climates, with blood pressure higher in the winter (low UV irradiation) than in the summer (high UV irradiation) 11, 12. Dark skin pigmentation, which affects an efficient UV light penetration 13, has also been reported to be associated with higher blood pressure 14, 15. Indeed, UV irradiation has been reported to lower blood pressure in patients with mild essential hypertension 16.
Numerous studies have shown that the serum level of 1,25(OH)2D3 is inversely associated with blood pressure in normotensive and hypertensive subjects 17-19. More interestingly, such inverse relationship has also been reported between circulating 1,25(OH)2D3 levels and plasma renin activity in patients with essential hypertension 20. Vitamin D supplement has been reported to be beneficial to the heart. In clinical trials, vitamin D treatment has been reported to reduce blood pressure in hypertensive or elderly patients 21, 22. In several cases, 1,25(OH)2D3 treatment has been shown to reduce the blood pressure, plasma renin activity, Ang II level and myocardial hypertrophy 23, 24.
Vitamin D as a negative endocrine regulator of the renin-angiotensin system
The clinical and epidemiological evidence suggests that vitamin D may regulate blood pressure via regulating the RAS. Based on the inverse relationship between serum 1,25(OH)2D3 levels and plasma renin activity found previously, we speculated that 1,25(OH)2D3 might be a negative endocrine regulator of renin production in vivo. If this hypothesis is correct, disruption of the vitamin D signaling pathway should lead to a deregulated stimulation of renin synthesis, whereas an increase in serum 1,25(OH)2D3 levels should lead to renin suppression.
To test this hypothesis, we examined VDR-null mutant mice, reasoning that renin expression should be increased in these mice due to the lack of VDR-mediated vitamin D signaling. Indeed, we found that both renin mRNA and protein levels in the kidney, as well as the plasma Ang II production, were drastically increased in VDR(-/-) mice, whereas the expression of angiotensinogen in the liver was the same as in wildtype mice. Thus, the increase in plasma Ang II appeared to be mainly due to an increase in renin activity. As a consequence of the aberrant RAS over-stimulation, VDR(-/-) mice developed hypertension, cardiac hypertrophy and an over-drinking behavior. Plasma and urinary aldosterone levels were also increased in VDR(-/-) mice. Cardiac hypertrophy, likely induced by Ang II, was reflected by a higher heart to body weight ratio and increased cardiac myocyte size in the ventricle revealed by histological analyses. Accompanying the cardiac hypertrophy, both cardiac mRNA expression and plasma concentration of atrial natriuretic peptide (ANP) were found to be increased in VDR(-/-) mice, presumably as a compensatory mechanism.
The high blood pressure, cardiac hypertrophy and increased water intake seen in VDR(-/-) mice could be corrected by treatment with captopril, an ACE inhibitor, or losartan, an Ang II AT1 receptor antagonist, confirming that over-stimulation of the RAS is indeed responsible for the abnormalities. Interestingly, captopril or losartan treatment resulted in a drastic up-regulation of renin expression in both wild-type and VDR(-/-) mice. We also showed that renin expression in both wild-type and VDR(-/-) mice was stimulated by dehydration, and suppressed by a high sodium diet. In all cases VDR(-/-) mice still maintained a significantly higher renin expression than wild-type mice. Therefore, despite a high basal renin synthesis, the regulatory mechanisms that control renin production, including the Ang II feedback inhibition and the volume- and salt-sensing mechanisms, are still intact in VDR(-/-) mice. Thus, the sustained renin up-regulation is mediated by other mechanisms.
VDR(-/-) mice develop hypocalcemia and secondary hyperparathyroidism 4, which may influence renin production and secretion. To address the contribution of serum calcium or parathyroid hormone (PTH) to renin up-regulation in VDR(-/-) mice, we found that renin up-regulation was already evident in 20-day old VDR(-/-) mice, before the hypocalcemia developed, and persistent in normocalcemic adult VDR(-/-) mice after their blood calcium was normalized through dietary intervention. The plasma Ang II level remained elevated in the normocalcemic state. On the other hand, renin expression was normal in Gcm2(-/-) mice 25, even though these mutant mice were as hypocalcemic as VDR(-/-) mice. In addition, renin expression was still elevated in VDR(-/-) mice whose alopecia was rescued by targeted expression of human VDR in the skin 26. These date strongly suggest that regulation of renin expression by 1,25(OH)2D3 is independent of calcium metabolism or alopecia. However, the contribution of PTH to the renin up-regulation in VDR(-/-) is less certain yet, because serum PTH starts to rise early in life before hypocalcemia develops and can not be completely normalized by dietary treatment, due to the lack of the VDR-mediated vitamin D inhibition of PTH biosynthesis 27.
We also tested the hypothesis using wild-type mice. We showed that in wild-type mice rendered vitamin D-deficient by dietary strontium treatment, which inhibits 1,25(OH)2D3 biosynthesis 28, renin expression in the kidney was up-regulated as in VDR(-/-) mice. On the other hand, in wild-type mice receiving several doses of 1,25(OH)2D3 injection renin expression was suppressed. Thus, the inhibitory role of 1,25(OH)2D3 in renin biosynthesis was confirmed in normal mice.
We further tested the hypothesis using As4.1 cells, a JG cell-like cell line that was derived from kidney tumors in SV40 T antigen transgenic mice and maintains a high level of renin synthesis 29. We found that 1,25(OH)2D3 drastically reduced renin mRNA expression in As4.1 cells transiently or stably transfected with human VDR cDNA. To elucidate the molecular mechanism whereby 1,25(OH)2D3 suppresses renin gene expression, the stably transfected cells were used to analyze the renin gene promoter by luciferase reporter assays. When the cells were transfected with a luciferase reporter plasmid containing a 4.1kb 5'-flanking sequence of the murine Ren-1c gene, 1,25(OH)2D3 treatment markedly reduced the promoter activity, confirming that 1,25(OH)2D3 directly and negatively regulates renin gene transcription by a VDR-mediated mechanism. Deletion analysis of the Ren-1c gene promoter has identified two short fragments, from -2720 to -2642 and -117 to +1, that are required to mediate the repression by 1,25(OH)2D3.
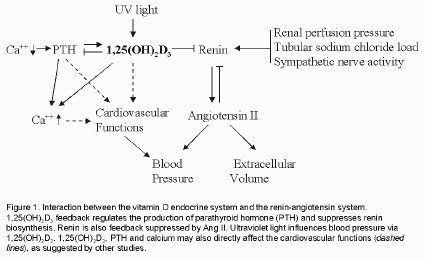
Conclusion
The discovery that 1,25(OH)2D3 suppresses renin gene expression helps explain, at least in part, the relationship between sunlight exposure, vitamin D and blood pressure observed previously. Figure 1 outlines the interaction between the vitamin D endocrine system and the RAS in the regulation of calcium, electrolytes, volume and blood pressure homeostasis. It is speculated that, under normal physiological conditions, in addition to maintaining the blood calcium concentration, 1,25(OH)2D3 may be one of the general "gate-keepers" to maintain an appropriate level of renin in the body, and/or may act as a counter-balance regulator to antagonize other renin-stimulating factors. The counter-balance of 1,25(OH)2D3 may be crucial to prevent the detrimental over-activation of the RAS. Therefore, vitamin D-deficiency may increase the risk of hypertension, and vitamin D supplement may be beneficial to the cardiovascular system.
This finding also raises a possibility to develop vitamin D analogs into renin inhibitors for therapeutic purposes. With a large number of low calcemic vitamin D analogs available, and some of them already approved for clinical uses such as treatment of secondary hyperparathyroidism 30, 31, such possibility is not unrealistic. As high-renin hypertension accounts for 10-20% of the patient population with essential hypertension, specific inhibitors for renin production are of significant therapeutic values. Low calcemic vitamin D analogs might open a new era for the long-sought therapeutic renin inhibitors and potentially offer a new class of anti-hypertensive drugs.
References
1. Haussler, M.R.,Whitfield, G.K., Haussler, C.A., Hsieh, J.-C., Thompson, P.D.,Selznick, S.H., Dominguez, C.E., and Jurutka, P.W. 1998. The nuclear vitamin D receptor: biological and molecular regulatory properties revealed. J. Bone and Mineral Research 13:325-349.
2. Yoshizawa, T., Handa, Y., Uematsu, Y., Takeda, S., Sekine, K., Yoshihara, Y., Kawakami, T., Arioka, K., Sato, H., Uchiyama, Y., et al. 1997. Mice lacking the vitamin D receptor exhibit impaired bone formation, uterine hypoplasia and growth retardation after weaning. Nature Genetics
16:391-396.
3. Panda, D.K., Miao, D., Tremblay, M.L., Sirois, J., Farookhi, R., Hendy, G.N., and Goltzman, D. 2001. Targeted ablation of the 25-hydroxyvitamin D1alpha -hydroxylase enzyme: evidence for skeletal, reproductive, and immune dysfunction. Proc Natl Acad Sci U S A 98:7498-7503.
4. Li, Y.C., Pirro, A.E.,Amling, M., Delling, G., Baron, R., Bronson, R., and Demay, M.B. 1997.Targeted ablation of the vitamin D receptor: an animal model of vitamin D-dependent rickets type II with alopecia. Proc Natl Acad Sci U S A 94:9831-9835.
5. Dardenne, O.,Prud'homme, J., Arabian, A., Glorieux, F.H., and St-Arnaud, R. 2001.Targeted inactivation of the 25-hydroxyvitamin D(3)-1(alpha)-hydroxylase gene (CYP27B1) creates an animal model of pseudovitamin D-deficiency rickets. Endocrinology 142:3135-3141.
6. Zinser, G., Packman,K., and Welsh, J. 2002. Vitamin D(3) receptor ablation alters mammary gland morphogenesis. Development 129:3067-3076.
7. Li, Y.C., Kong, J., Wei, M., Chen,
Z.F., Liu, S.Q., and Cao, L.P. 2002. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest 110:229-238.
8. Kong, J., and Li, Y.C. 2003. Effect of Angiotensin II Type I Receptor Antagonist and Angiotensin-Converting Enzyme Inhibitor on Vitamin D Receptor Null Mice. Am J Physiol Regul Integr Comp Physiol 285:R255-R261.
9. Rostand, S.G. 1997. Ultraviolet light may contribute to geographic and racial blood pressure differences. Hypertension 30:150-156.
10. He, J., Klag, M.J., Wu, Z., and Whelton, P.K. 1995. Stroke in the People's Republic of China. I. Geographic variations in incidence and risk factors. Stroke
26:2222-2227.
11. Kunes, J., Tremblay, J., Bellavance, F., and Hamet, P. 1991. Influence of environmental temperature on the blood pressure of hypertensive patients in Montreal. Am J Hypertens 4:422-426.
12. Woodhouse, P.R., Khaw, K.T., and Plummer, M. 1993. Seasonal variation of blood pressure and its relationship to ambient temperature in an elderly population. J Hypertens 11:1267-1274.
13. Holick, M.F. 1987. Photosynthesis of vitamin D in the skin:
effect of environmental and life-style variables. Fed Proc 46:1876-1882.
14. Harburg, E., Gleibermann, L., Roeper, P., Schork, M.A., and Schull, W.J. 1978. Skin color, ethnicity, and blood pressure
I: Detroit blacks. Am J Public Health 68:1177-1183.
15. Klag, M.J., Whelton, P.K., Coresh, J., Grim, C.E., and Kuller, L.H. 1991. The association of skin color with blood
pressure in US blacks with low socioeconomic status. Jama265:599-602.
16. Krause, R., Buhring, M.,Hopfenmuller, W., Holick, M.F., and Sharma, A.M. 1998. Ultraviolet B and blood pressure. Lancet352:709-710.
17.Kristal-Boneh, E., Froom, P., Harari, G., and Ribak, J. 1997. Association of calcitriol and blood pressure in normotensive men. Hypertension 30:1289-1294.
18. Lind, L., Hanni, A., Lithell, H., Hvarfner, A., Sorensen, O.H., and Ljunghall, S. 1995. Vitamin D is related to blood pressure and other cardiovascular risk factors in middle-aged men. Am J Hypertens 8:894-901.
19. Burgess, E.D., Hawkins, R.G., and Watanabe, M. 1990. Interaction of 1,25-dihydroxyvitamin D and plasma renin activity in high renin essential hypertension. Am J Hypertens 3:903-905.
20. Resnick, L.M., Muller, F.B., and Laragh, J.H. 1986. Calcium-regulating hormones in essential hypertension. Relation to plasma renin activity and sodium metabolism. Ann Intern Med 105:649-654.
21. Lind, L., Wengle, B., Wide, L., and Ljunghall, S. 1989. Reduction of blood pressure during long-term treatment with active vitamin D (alphacalcidol) is dependent on plasma renin activity and calcium status. A double-blind, placebo-controlled study. Am J Hypertens 2:20-25.
22. Pfeifer, M., Begerow, B., Minne, H.W., Nachtigall, D., and Hansen, C. 2001. Effects of a short-term vitamin D(3) and calcium supplementation on blood pressure and parathyroid hormone levels in elderly women. J Clin Endocrinol Metab 86:1633-1637.
23. Kimura, Y., Kawamura, M., Owada, M., Oshima, T., Murooka, M., Fujiwara, T., and Hiramori, K. 1999. Effectiveness of 1,25-dihydroxyvitamin D supplementation on blood pressure reduction in a pseudohypoparathyroidism patient with high renin activity. Intern Med 38:31-35.
24. Park, C.W., Oh, Y.S., Shin, Y.S., Kim, C.M., Kim,
Y.S., Kim, S.Y., Choi, E.J., Chang, Y.S., and Bang, B.K. 1999. Intravenous calcitriol regresses myocardial hypertrophy in
hemodialysis patients with secondary hyperparathyroidism. Am J Kidney Dis 33:73-81.
25. Gunther, T., Chen, Z.F., Kim, J., Priemel, M., Rueger, J.M., Amling, M., Moseley, J.M., Martin, T.J., Anderson, D.J., and Karsenty, G. 2000. Genetic ablation of parathyroid glands reveals another source of parathyroid hormone. Nature 406:199-203.
26. Kong, J., Li, X.J., Gavin, D., Jiang, Y., and Li, Y.C. 2002. Targeted expression of human vitamin D receptor in the skin promotes the initiation of postnatal hair follicular cycle and rescue the alopecia in vitamin D receptor null mice. J. Invest. Dermatol. 118:631-638.
27. Silver, J., Naveh-many, T., Mayer, H., Schmeizer, H.J., and Popovtzer, M.M. 1986. Regulation by vitamin D metabolites of parathyroid hormone gene transcription in vivo in the rat. J. Clin. Invest. 78:1296-1301.
28. Omdahl, J.L., and DeLuca, H.F. 1971. Strontium induced rickets: metabolic basis. Science 174:949-951.
29. Sigmund,C.D., Okuyama, K., Ingelfinger, J., Jones, C.A., Mullins, J.J., Kane, C., Kim, U., Wu, C.Z., Kenny, L., Rustum, Y., et al. 1990. Isolation and characterization of renin-expressing cell lines from transgenic mice containing a renin-promoter viral oncogene fusion construct. J Biol Chem 265:19916-19922.
30. Brown, A.J., Dusso, A.S., and Slatopolsky, E. 2002. Vitamin D analogues for secondary hyperparathyroidism. Nephrol Dial Transplant 17 Suppl 10:10-19.
31. Malluche, H.H., Mawad, H., and Koszewski, N.J. 2002. Update on vitamin D and its newer analogues: actions and rationale for treatment in chronic renal failure. Kidney Int 62:367-374. |
