
|
Paneles de Discussión
Paneais de Discussio |
"EARLY DIAGNOSIS OF POLYOMA VIRUS ALLOGRAFT NEPHROPATHY"Cinthia B. Drachenberg, M.D., John C. Papadimitriou, M.D., Ph.D. and Emilio Ramos M.D.Departments of Pathology and Medicine. University of Maryland School of Medicine. Baltimore. USAcdrac001@umaryland.eduThe BK virus was isolated in 1971 from the urine of a renal transplant patient. After 1995 a marked increase in the incidence of BK allograft nephritis has occurred. Currently BK-associated nephropathy (BKAN) is considered an important cause of allograft dysfunction and failure. BK viral excretion in urine occurs mostly within the first 3 months renal transplantation, but can also appear late. BKAN has been identified in much fewer (1-8%) renal transplant recipients 1. The increased recognition of BK allograft nephropathy in part results from the advent of newer diagnostic techniques. On the other hand, the increased incidence and virulence of the disease has coincided with the development of more potent immunosuppressive agents. These include Tacrolimus, Cyclosporine, and Mycophenolate Mofetil, in addition to the antilymphocyte antibodies used for induction or treatment of renal allograft rejection. Viral replication appears to be also stimulated by an alloimmune response caused by, or associated with graft rejection, inflammation and tissue injury 2. There is general agreement that an increased immunosuppressive burden is a risk factor for the infection, and in the absence of an effective antiviral treatment, reduction of immunosuppression is warranted to treat the infection 3.
For the diagnosis of BKAN, BK viral cytopathic changes are to be found in a renal biopsy. The nature of the viral changes have to be confirmed with immunohistochemical stains, insitu-hybridization, PCR, and/or electron microscopy 4. The primary infection and BK viral reactivation (with or without renal dysfunction) are manifested by viruria. The infected epithelial cells that exfoliate from the renal tubules and urothelium ("decoy cells") can be identified with routine cytological evaluation of voided urine. Qualitative urine PCR is a much more sensitive method to identify viruria. This molecular method is exquisitely sensitive and identifies very small amounts of BK virions that can be excreted in the urine even of normal individuals. Because qualitative PCR is too sensitive, quantitative urine PCR measurements are the most potentially useful and research is being done to standardize this method. Twenty to 30% of renal transplant recipients can have decoy cells in urine at some point during the post-transplantation course, indicating BK virus replication in the urinary tract. In our experience, persistence of decoy cells in urine over several months has coincided or preceded the diagnosis of BKAN in all our patients. In one patient, positive urine cytologies preceded the development of nephritis for up to 15 months. On the other hand, viruria occurring in association with non BK-related tubular damage (i.e. severe acute rejection, acute tubular necrosis, calcineurin inhibitor toxicity, pyelonephritis, etc.) is usually transient 5. Measurements of BK viral load in plasma has been demonstrated to have good correlation with BKAN. Although the number of patients studied are relatively small, it appears that a viral load of >10,000 ml plasma is a good indicator of BKAN 2. Despite its limitations, evaluation of urine cytology continues to be the most simple and inexpensive method for screening purposes. Consequently, at the University of Maryland Hospital we routinely perform cytological evaluation of urine in transplant patients as described in Figure 1. Figure 1
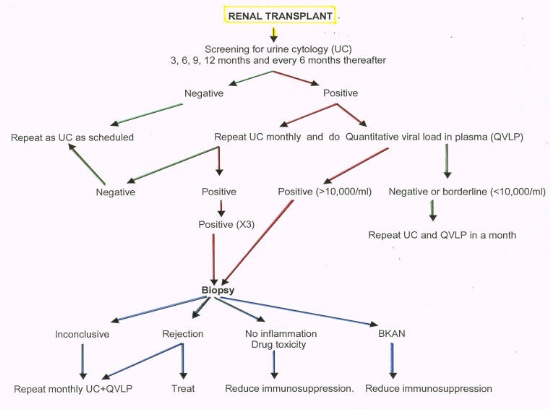
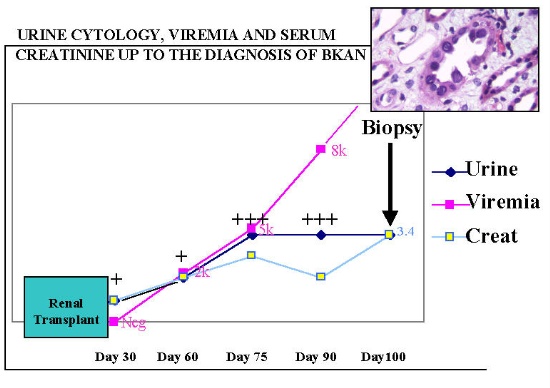
A positive urine cytology is an indication of measurement of BK viral load in plasma. If the urine cytology is repeatedly positive a renal biopsy is indicated. The determination of a therapeutic intervention depends on a combination of urine cytology, renal biopsy and viremia. Due to sampling errors, a renal biopsy may be falsely negative in the presence of a significant viremia and viruria 3. However, it is necessary to always rule out other pathological process (most importantly acute rejection), before any adjustment of immunosuppression is made. We have seen the best results when reduction in immunosuppression results in the use of prednisone and a single immunosuppressant drug (FK506, CSA , sirolimus or MMF). Early diagnosis of BKAN with a combination of urine cytology, measurement of viremia and renal biopsy is exemplified in Figure 2. A 52 year old female underwent a cadaveric renal transplant with initial good function. Screening urine cytology indicated the presence of infected cells in urine (decoy cells) at 30 days post-transplantation. Urine cytology persisted to be positive as the viremia increased and the renal function deteriorated. A biopsy at day 100 showed early polyoma virus nephropathy. The picture shows a renal tubule lined by infected tubular cells, which appear large and hyperchromatic. Figure 2
BK allograft nephropathy is an important cause of premature kidney graft loss at the University of Maryland transplant program. One third of the patients diagnosed with BKAN have already lost graft function, and a third of the remaining patients have creatinines over 3 mg/dl (ref). After reduction of immunosuppression, the course of BKAN in most patients falls into one of the following categories.
2) Persistent BKAN complicated with cycles of acute allograft rejection. Approximately 10% of patients followed this course and eventually lost graft function. 3) Persistence of viral replication with continuous associated tubular damage and accelerated rate of graft sclerosis in comparison to patients without BKAN. Graft loss can be rapid within few months or more protracted. This pattern was observed in the majority of patients with BKAN 3. We have previously speculated that poor results may be associated with late intervention when a significant amount of irreversible tissue damage has been established 6. A comparison between patients diagnosed prospectively versus patients biopsied when the renal function is already impaired has shown a better renal function in the first group. In patients diagnosed with early biopsy triggered by persistence of BK virus infected cells in urine, there is a higher rate of conversion from positive to negative urine cytology as well as stabilization of renal function with lower creatinines 7. Renal transplant patients should be screened for evidence of polyoma virus reactivation in order to prevent irreversible graft damage and accelerated graft loss.
References:
2. Hirsch HH, Knowles W, Dickenmann M, Passweg J, Klimkait T, Mihatsch MJ, Steiger J: Prospective study of polyomavirus type BK replication and nephropathy in renal-transplant recipients. N Engl J Med 2002;347: 488-496. 3. Ramos E, Drachenberg CB, Papadimitriou JC, Hamze O, Fink JC, Klassen DK, Drachenberg RC, Wiland A, Wali R, Cangro CB, Schweitzer E, Bartlett ST, Weir MR: Clinical course of polyoma virus nephropathy in 67 renal transplant patients. J Am Soc Nephrol 2002;13:2145-2151. 4. Randhawa PS, Demetris AJ: Nephropahty due to polyomavirus type BK. N Engl J Med 2002;342:1361-1363. 5. Drachenberg RC, Drachenberg CB, Papadimitriou JC et al.: Morphological spectrum of polyoma virus disease in renal allograft: Diagnostic accuracy of urine cytology. Am J Transpl 2001;1:373-81. 6. Ramos E, Drachenberg CB, Papadimitriou JC et al: Impact of prospective urine cytology on graft function with earlier diagnosis of polyoma virus nephropathy. J Am Soc Nephrol 2002;13:378A 7. Ramos E, Drachenberg CB, Portocarrero M et al.: BK virus nephropathy diagnosis and treatment: Experience at the University of Maryland Renal Transplant Program. In: JM Cecka, PI Terasaki eds. UCLA Immunogenetics Center, Los Angeles CA. Clinical Transplants 2002, 2003;143-153. |