
|
Paneles de Discussión
Paneais de Discussio |
Glomerulopathies with Organized DepositsSamy S. Iskandar, MBBCh, PhD.Professor of Pathology. Wake Forest University School of Medicine.
iskandar@wfubmc.edu |
 |
 |
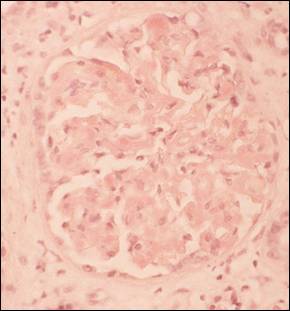
Electron microscopy reveals the randomly disposed 7-15 nm fibrils in the mesangium and in more advanced cases the glomerular basement membrane (GBM) is infiltrated, often in the peculiar pattern illustrated below, which is responsible for the appearance of spicules in silver impregnated section of the paraffin embedded material (Figure 3). Although some standard pathology textbooks give a range of 7 -10 nm, our own experience is more in line the extended range. There is some overlap with fibrillary glomerulonephritis (FGN). Therefore, although this is the most specific examination modality, Congo-red negativity has to be demonstrated to avoid misdiagnosing fibrillary glomerulonephritis as amyloidosis.
Fibrillary Glomerulonephritis:
Is an immune complex-mediated glomerulopathy characterized by the identification of randomly disposed Congo-red negative fibrils by electron microscopy. In multiple large biopsy series the incidence has been reported to be 0.6% to 1% of native kidney biopsies2-5.
Diagnosis:
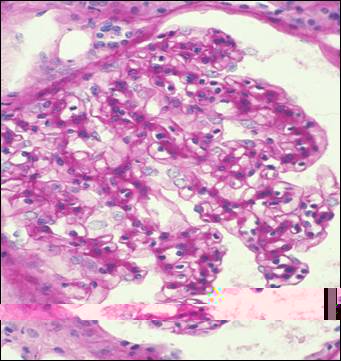
On bright field microscopy the pattern is highly variable and ranges from mesangial proliferative to membranoproliferative. But I have seen a pattern of focal segmental sclerosis in an occasional case. Crescents have been reported in as much as 31% of cases. In all but the most subtle cases, the mesangial matrix will be clearly highlighted by its intense staining with the PAS method (Figure 4). Our experience with this regard is contrary to that of the Columbia College of Physicians and Surgeons group who found the tinctorial properties of FGN to be the same as those of amyloid infiltrates. By definition the Congo-red stain is negative
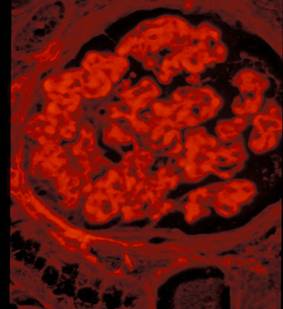
The diagnosis can be anticipated at the time of immunofluorescence study. Deposits typically show reactivity with IgG, particularly the IgG4 subclass, and C3. Most helpful is the quality of the fluorescence. Although the fluorescence signal is strong, it typically has a subdued quality to it, which some describe as smudged for lack of better term (Figure 5). A pattern of glomerular basement membrane staining simulating the linear fluorescence of anti-GBM disease has been reported; another pitfall which can probably be avoided by paying attention to the quality of the immunofluorescence signal—although this is unlikely to be a problem in the global clinical and morphological context. When subclass of IgG is looked for there is a clear predominance of staining with anti-IgG4. Dr. D’Agatti’s group in New York 4 found reactivity for both IgG1 and IgG4 in 15 of 19 case tested with the remaining four equally divided between exclusive IgG1 and IgG4 reactivity.

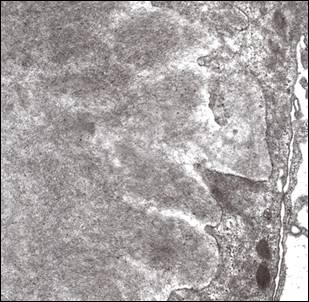
On electron microscopic examination, randomly disposed fibrils produce a felt work which resembles the pattern seen in amyloidosis, hence the initial nomenclature of Congo red-negative amyloidosis-like glomerulopathy. Again the fibrils are seen in the mesangium but can obliterate the GBM producing a spiculated pattern which can simulate that illustrated in Figure 3. Fibrils are roughly 15-30 nm in width i.e. approximately double the size of amyloid fibrils (Figure 6). However, because of the overlap with amyloidosis at the lower end of this range, Congo-red negativity is mandatory to confidently discern between the two conditions. A pitfall for the novice is misinterpretation of the sometimes particularly prominent fibrillar appearance of the mesangial matrix in conditions associated with glomerular scarring6 as FGN (Figure 7). This can be avoided if one pays attention to the substructural details which shows bundles often with a serpentine appearance rather than the random pattern seen in bona fide FGN. Also one has to remember that FGN has distinctive immunofluorescence features as detailed above; whereas in most cases presenting a prominent fibrillar appearance to the mesangial matrix, the immunofluorescence findings are non-specific.
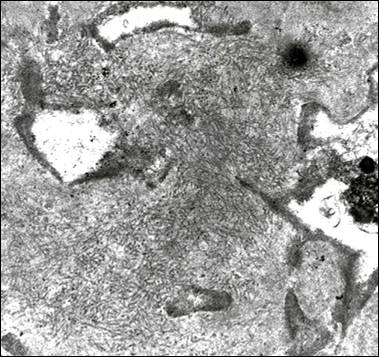
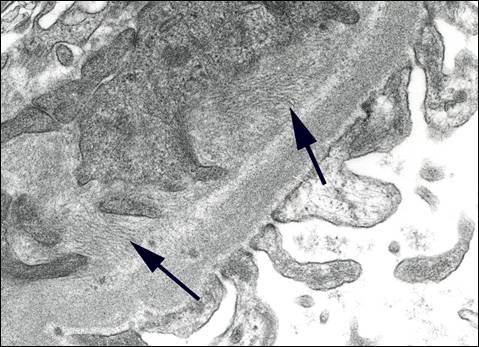
In contrast to amyloidosis which is usually a systemic disease, fibrillary glomerulonephritis is a kidney limited lesion. In the overwhelming majority of cases the fibrils are present in glomeruli only. Occasionally, fibrils have also been found in tubular basement membranes. Rare reports of pulmonary involvement7 should be viewed with some reservation since in conditions associated with pulmonary fibrosis; matrical fibrils can introduce a confounding diagnostic element8.
Immunotactoid Glomerulopathy (ITG):
We and other investigators believe that the term should be reserved to a form of glomerulopathy characterized by the presence of microtubules arranged parallel bundles and showing a central lumen when examined at the magnifications used for routine diagnostic purposes. This issue will be addressed further below. The entity has an incidence that is one tenth that of FGN. Interestingly though, a recent study comparing the two entities included 14 cases of ITG and only 9 cases of FGN5.
Diagnosis:
With bright field microscopy, the features are not significantly different from those of FGN.
Immunofluorescence is similar to that of FGN except that when the subclass of IgG deposits has been looked into there was a spread of IgG1, IgG2 and IgG3 but interestingly no cases stained with anti-IgG4.
On electron microscopy the microtubules as indicated above are arranged in parallel arrays. And have a distinct lumen. Their diameter is typically > 30 nm and can be as high as 70 nm. The recent study of Bridoux et al. cited above found a smaller range of 9 - 45 nm. There has been an ongoing polemic about whether or not one should distinguish FGN and ITG. Some authors favor grouping all cases under the label IT glomerulopathy9. There has been agreement though amongst the authors of the several largest series published to date that there is ground to distinguish the two entities, both on the basis of their distinctive ultrastructural features and the fact that cases of IT glomerulopathy show a high association with monoclonal gammopathy, and hematological malignancy2-5,10 it was found that the incidence of lymphoproliferative neoplasia, if one included patients with serum or urine paraproteins, was 1% to 5% in cases pf FGN and between 33% and 45% in cases of ITG. The different proportions being related to whether one assigned a case to a particular category based on the fibrils/microtubules’ diameter (<30 nm for FGN and >30 nm for ITG) or their arrangement (random in FGN and focally organized in ITG).
Cryoglobulinemic Glomerulopathy:
Organized deposits can be seen in types in types I and II (pure monoclonal vs. monoclonal IgM to polyclonal IgG, respectively).
Diagnosis:
By routine bright field microscopy there is most frequently a membranoproliferative (mesangiocapillary)-like pattern with the hypercellularity attributable to mesangial cell and monocytes/macrophages. Most helpful is identification of the characteristic hyaline thrombi, which can be quite focal and segmental and may require careful search.
Electron microscopy, if one is fortunate enough to sample the right field, shows subendothelial and/or luminal deposits of electron-dense material with short curved microtubules which may show a pattern of focal organization into small stacks (Figure 8). These are the equivalents of the hyaline thrombi seen by bright field microscopy. In type III cryoglobulinemia the deposits show only a vague microtubular substructure.
Immunofluorescence usually reflects the clonality of the cryoglobulins involved. The pattern is usually not that of a classic idiopathic membranoproliferative glomerulonephritis.
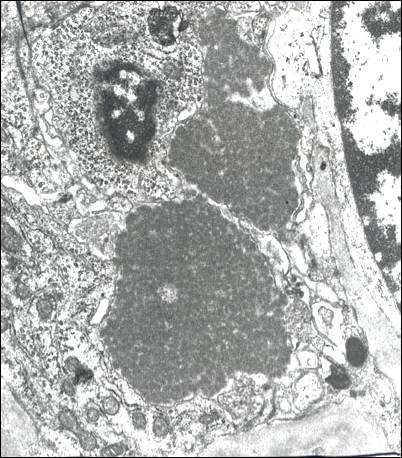
Fibronectin Glomerulopathy:
Is a recently described entity in several families in different parts of the world with patients presenting with proteinuria often of nephrotic level and microhematuria.
Diagnosis:
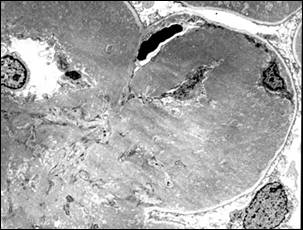
Bright field microscopy shows a pattern of accentuation of the lobular architecture of the glomerular tuft due to mesangial expansion (Figure 9A, courtesy of Dr. Arthur Abt, Penn State College of Medicine). There is also capillary wall thickening, but no peripheral mesangial interposition. The expanded mesangium and thickened capillary walls stain strongly with the PAS method but are not argyrophilic.
Immunofluorescence is noncontributory. The diagnosis is provided by immunohistochemical demonstration of strong reactivity with antibodies to serum fibronectin in the same distribution as the PAS staining material.
Electron microscopy shows extensive electron-dense material in the mesangium and capillary wall (Figure 9B, courtesy of Dr. Arthur Abt, Penn State College of Medicine). There appears to be differences in different kindred, in some the deposits are mostly granular whereas others show a feltwork of fibrils 14 to 16 nm in diameter11,12.
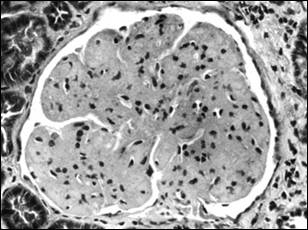 |
 |
Collagenofibrotic Glomerulopathy:
Characterized by abundant deposition of type III collagen.
Diagnosis:
The bright field microscopy picture is that of accentuation of the lobular architecture of the glomeruli. The differential diagnosis is therefore that of MPGN and its mimics13.
Immunofluorescence microscopy shows focal segmental staining with IgG, IgM, and C1q along peripheral capillary walls.
Electron microscopy shows extensive deposits of type III collagen in an expanded subendothelial space.
Acknowledgements: I wish to acknowledge the invaluable assistance of Dr. Jerald Winter whose experience with computer technology made this presentation possible as well as the technical assistance of Mr. Scott Evans, Mr. Kenneth Grant, Ms. Belinda Dawson and Ms. Paula Moore
REFERENCES
1. Iskandar SS, Herrera GA: Glomerulopathies with organized deposits. Semin Diagn Pathol 2002, 19:116-132
2. Fogo A, Qureshi N, Horn RG: Morphologic and clinical features of fibrillary glomerulonephritis versus immunotactoid glomerulopathy [see comments] [published erratum appears in Am J Kidney Dis 1993 Nov;22(5):757]. Am J Kidney Dis 1993, 22:367-377
3. Iskandar SS, Falk RJ, Jennette JC: Clinical and pathologic features of fibrillary glomerulonephritis. Kidney Int 1992, 42:1401-1407
4. Rosenstock JL, Markowitz GS, Valeri AM, Sacchi G, Appel GB, D'Agati VD: Fibrillary and immunotactoid glomerulonephritis: Distinct entities with different clinical and pathologic features. Kidney Int 2003, 63:1450-1461
5. Bridoux F, Hugue V, Coldefey O, Goujon J-M, Bauwens M, Sechet A, Preud'homme J-L, Touchard G: Fibrillary glomerulonephritis and immunotactoid (microtubular) glomerulopathy are associated with distinct immunological features. Kidney Int 2002, 62:1764-1775
6. Kronz JD, Neu AM, Nadasdy T: When noncongophilic glomerular fibrils do not represent fibrillary glomerulonephritis: nonspecific mesangial fibrils in sclerosing glomeruli. Clin Nephrol 1998, 50:218-223
7. Masson RG, Rennke HG, Gottlieb MN: Pulmonary hemorrhage in a patient with fibrillary glomerulonephritis. N Engl J Med 1992, 326:36-39.
8. Jennette JC, Iskandar SS, Falk RJ: Fibrillary glomerulonephritis. Renal Pathology with Clinical and Functional Correlations. Edited by Tisher CC, Brenner BM. Philadelphia, J. B. Lippincott Company, 1994, pp 553-563
9. Korbet SM, Schwartz MM, Lewis EJ: The fibrillary glomerulopathies. Am J Kidney Dis 1994, 23:751-765.
10. Devaney K, Sabnis SG, Antonovych TT: Nonamyloidotic fibrillary glomerulopathy, immunotactoid glomerulopathy, and the differential diagnosis of filamentous glomerulopathies. Mod Pathol 1991, 4:36-45.
11. Strřm EH, Banfi G, Krapf R, Abt AB, Mazzucco G, Monga G, Gloor F, Neuweiler J, Riess R, Stosiek P, Hebert LA, Sedmal DD, Gudat F, Mihatsch MJ: Glomerulopathy associated with predominant fibronectin deposits: a newly recognized hereditary disease. Kidney Int 1995, 48:163-170.
12. Gemperle O, Neuweiler J, Reutter FW, Hildebrandt F, Krapf R: Familial glomerulopathy with giant fibrillar (fibronectin-positive) deposits: 15-year follow-up in a large kindred. Am J Kidney Dis 1996, 28:668-675.
13. Gubler MC, Dommergues JP, Foulard M, Bensman A, Leroy JP, Broyer M, Habib R: Collagen type III glomerulopathy: a new type of hereditary nephropathy. Pediatr Nephrol 1993, 7:354-360