
|
Paneles de Discussión
Paneais de Discussio |
POLYOMAVIRUS INFECTIONS IN KIDNEY TRANSPLANT RECIPIENTSParmjeet Randhawa MD.Departments of Pathology. University of Pittsburgh School of Medicine. Pittsburgh, Pennsylvania. USArandhawapa@upmc.edu
Polyomaviruses are 45nm sized particle with a 5 kb genome 1, 2 The viral genome averages 5 kilobases in length, and is comprised of double-stranded circular supercoiled DNA. The viral genome is typically arranged in three general regions: non-coding control region (NCCR), the early coding region coding for the small and large T antigens, and the late coding region coding for the viral capsid proteins (VP-1, VP-2, VP-3) and agnoprotein. The direction of early and late transcription is divergent, with opposite DNA strands participating in these processes3. The NCCR contains (a) the origin of replication (ori), and (b) regulatory regions containing enhancer elements that are important activators of viral transcription 4. Naturally occurring BKV and JCV strains in the kidney and urine usually have an archetypal regulatory region, although this in not an invariable rule 5, 6. By contrast, JCV found in the brain tissue of patients with progressive multifocal leucoencephalopathy (PML) usually shows a variety of genetic mutations & rearrangements 7-9. There is in-vitro evidence that NCCR variants determine host cell permissivity and rate of viral replication 9, 10. Hence, genetic rearrangements are believed to be critical in permitting viral transcription in the brain, which ultimately culminates in the pathologic lesions of progressive multifocal leucoencephalopathy. Such rearrangements have been described in isolated case reports of immunodeficient individuals with BKV infection, but are present only in a small proportion of patients with BKV nephropathy. The T antigens bind to tumor suppressor proteins Rb and p53 and stimulate host cell entry into the cell cycle 13, 14. This observation provides a theoretical basis for multiple lines of accumulating evidence that PV may be carcinogenic in man: (a). PV can transform fibroblast cell lines and is a well established carcinogen in experimental animals 3, 15, (b) BKV DNA has been found in renal cell carcinoma and urothelial carcinoma 16-18, (c) Glial tumors contain JCV DNA, and infectious JCV has been recovered from a patient with a choroid plexus tumor3, (d) SV40 DNA has been repeatedly found in human tumors (see section B.2.), and (e) mesothelioma tissue has been found to contain functionally active SV40 T-antigen capable of binding to the tumor suppressor protein p53 13, 19-21. Nonetheless, the idea that PV can cause human cancer remains unproven because of the ubiquitous latency of this group of viruses. The viral capsid proteins VP-1, VP-2, and VP-3 are structural proteins required for the assembly of complete virions. The viral capsid coding regions display considerable genetic heterogeneity, and this feature has been used to divide BKV into distinct genotypes I, II, III, and IV 22-26, and JCV into Types 1, 2A, 2B, and 3-8 27-33. In mice, specific mutations in the viral capsid protein VP-1 region have been associated with increased viral pathogenicity 34, 35. Whether similar considerations apply to human patients with BKV associated renal allograft interstitial nephritis is unknown. The existence of potential relationships between viral genotype and clinical virulence is illustrated by the observation that progressive multifocal leukoencephalopathy is associated primarily with JCV Type 2B infection. Agnoprotein protein localizes primarily to the cytoplasmic and perinuclear regions of the host cell. This distribution has led to the suggestion that agnoprotein may promote virion release from cell 36. Other proposed roles for this protein include participation in host cell lysis, enhanced nuclear localization of viral capsid protein VP-1, and help in viral capsid assembly 37. Cultured cells infected with agnogene mutants show a 17-100 fold reduction in virion burst size 38. JCV agnoprotein has recently been localized in medulloblstoma 39. No data is currently available on the occurrence of mutant agnoproteins in human disease. An overview of the clinical aspects of PV infections in man follows. The emphasis is on BKV, which is currently believed to be the principal etiologic agent of renal disease in immunosuppressed individuals. However, simian polyoma virus SV40 and JCV will also be discussed briefly.
SV40 infections in man: SV40 is a simian virus, which causes a disease resembling progressive multifocal leucoencephalopathy in immunocompromised monkeys. The use of live vaccines prepared in monkey cells led to about 10-30 million human beings getting infected by this virus between 1954 and 1963 3, 40. There are now increasing reports documenting authentic SV40 sequences in a variety of human neoplasms, including mesothelioma, choroid plexus papilloma, ependymoma, non-Hodgkin lymphomas, and osteogenic sarcomas20, 41-46. Viral sequences have been found in leucocytes of healthy blood donors, and native kidneys with focal segmental sclerosis, minimal change disease, and membranous glomerulopathy 47. Simian polyoma virus SV40 sequences have recently been documented in healthy kidney transplant recipients 48, and in the blood and urine of patients with PVAN . A pathogenic role for SV40 in viral nephropathy has not yet been established, but this is an area which needs more systematic investigation. JCV infections in man: JCV was initially isolated from a patient with PML, and can be found in up to 75% of blood & 92% of cerebrospinal fluid samples obtained from these patients 49-52. Subsequent studies showed widespread serologic evidence of infection in human populations 53. Following primary infection, the virus becomes latent in the kidney and B-lymphocytes 54-56. JCV can be excreted with significant frequency in the urine of healthy individuals 29, 32, 57, 58. Unlike BKV, some studies have failed to find a correlation between the frequency of JCV viruria and the degree of immunosuppression 59-61. However, PML occurs primarily in the setting of immune deficiency, such as in patients with AIDS, solid organ transplantation, lymphoproliferative disorders, neoplastic diseases, myeloproliferative syndromes, and sarcoidosis 53, 62, 63. Although type 2 is the most common genotype associated with PML, type 1 cases have also been reported 64. It is not certain whether JC virus can cause renal disease. However, we have found JCV DNA to be present in 7/19 (37%) renal allograft biopsies with BKV associated PVAN 65. The frequency of JCV in allograft biopsies appears to be higher than that observed in native and allograft kidneys without polyomavirus nephropathy (PVAN), where it was found in 0/19 (0%) samples. Studies of JCV gene expression and genomic mutations in allograft kidney tissue are needed further evaluate a potential role of JCV in causing renal allograft dysfunction in man.
BKV was first isolated in 1970, & subsequent studies documented a world-wide seroprevalence rate of 60-80% 1, 2, 66. Initial infections usually occur in childhood putatively via the respiratory route, followed by viral latency in the urogenital tract. BKV reactivation with urinary shedding of infected urothelial cells is recognized to occur in 10-60% of renal transplant recipients. Receipt of a kidney from a seropositive donor is a risk factor for lytic infection regardless of the serologic status of the recipient at the time of transplantation 1, 2. Most early studies found only sporadic cases of graft dysfunction associated with viral activation, and did not report any biopsy findings 67-69. BKV-associated interstitial nephritis associated with serious graft dysfunction was initially described in the setting of congenital immunodeficiency and AIDS 70, 71. More recent studies have shown that BK virus can cause PVAN in up to 8% of renal allografts 17, 72-79. Typically, a diagnosis of BKV associated PVAN is made by recognition of viral inclusions in renal tubular and glomerular epithelial cells at allograft biopsy. Viral inclusions are associated with variable mononuclear interstitial infiltrates and focal tubulitis that closely resembles acute rejection. It is not known if this resemblance reflects common mechanisms of T-cell influx, or the concurrent presence of viral infection and true acute rejection. The relationship between PV infection and acute as well as chronic graft rejection remains to be evaluated in a systematic fashion, as has been done for cytomegalovirus 80, 81 Conceivably, PV infection could precipitate acute rejection via upregulation of MHC class I or class II antigen expression, or the release of other pro-inflammatory cytokines. However, the cytokine milieu associated with human PVAN has not been specifically investigated. Conversely, experimental evidence suggests that tubular injury associated with acute cellular rejection could secondarily increase PV replication 82. In approximately 30% of the biopsies with PVAN, a significant inflammatory infiltrate of polymorphs is also present. Prominence of plasma cells and changes resembling acute tubular necrosis are other histomorphologic features described in this clinical setting. Although, a definitive diagnosis of PVAN currently requires a biopsy, there is considerable interest in developing less invasive diagnostic methods. It has been found that on urine cytology, most cases with detectable viral inclusions are asymptomatic, but presence of >10 inclusion bearing cells with an accompanying inflammatory response identifies all cases of PVAN 76. Urine viral load > 1E+07 copies/ml and plasma viral load > 1E+04 copies/ml tends to correlate with histologic nephropathy, but a biopsy is needed for definitive diagnosis. Clinical management of BKV renal allograft nephropathy remains a significant challenge. Even when biopsies show tubulitis suggesting the possibility of underlying rejection, there is little or only transient response to corticosteroids in most cases. Conversely, reducing immunosuppression decreases the viral load, but increases the risk of rejection. This therapeutic dilemma results in an unfavorable graft outcome, which is unlikely to improve till clinically effective anti-polyomavirus drugs are developed. No clinically proven anti-PV drugs are currently available for the management of PVAN. However, we have had encouraging results with cidofovir, which typically results in clearance of viremia, and several log reduction in the urinary titers of virus 86. Controlled trials need to be performed to better substantiate the efficacy of this drug. Less nephrotoxic antiviral compounds also need to be evaluated for potential clinical use. For example, retinoic acid derivatives, nalidixic acid and oxolinic acid suppress replication and cytopathic effect of BKV in vitro87, 88. Topoisomerase inhibitors block JCV replication in fetal glial cells and have had variable success in the therapy of patients with PML 89. Lefunomide is another drug that has been recently used in BKV nephropathy 90. 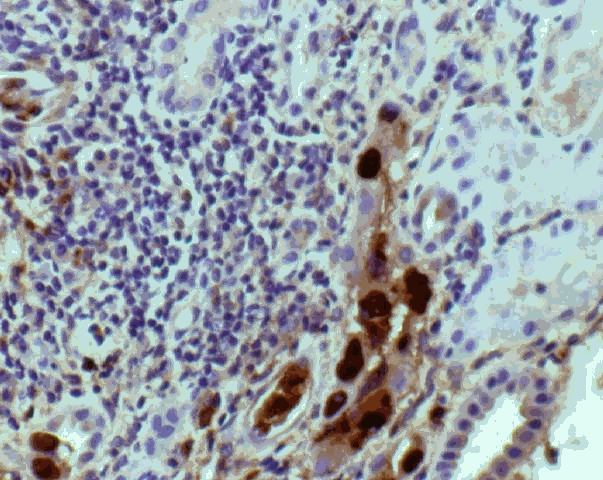 Figure 1: References
2. Shah KV. Human polyomavirus BKV and renal disease. Nephrology Dialysis Transplantation 2000; 15 (6): 754. 3. Lednicky JA, Butel JS. Polyomaviruses and human tumors: a brief review of current concepts and interpretations. Front Biosci 1999; 4: D153. 4. Moens U, Johansen T, Johnsen JI, Seternes OM, Traavik T. Noncoding control region of naturally occurring BK virus variants: sequence comparison and functional analysis. Virus Genes 1995; 10 (3): 261. 5. Dorries K, Vogel E, Gunther S, Czub S. Infection of human polyomaviruses JC and BK in peripheral blood leukocytes from immunocompetent individuals. Virology 1994; 198 (1): 59. 6. Sundsfjord A, Johansen T, Flaegstad T, et al. At least two types of control regions can be found among naturally occurring BK virus strains. Journal of Virology 1990; 64 (8): 3864. 7. Newman JT, Frisque RJ. Detection of archetype and rearranged variants of JC virus in multiple tissues from a pediatric PML patient. Journal of Medical Virology 1997; 52 (3): 243. 8. Loeber G, Dorries K. DNA rearrangements in organ-specific variants of polyomavirus JC strain GS. J Virol 1988; 62 (5): 1730. 9. Johnsen JI, Seternes OM, Johansen T, Moens U, Mantyjarvi R, Traavik T. Subpopulations of non-coding control region variants within a cell culture-passaged stock of BK virus: sequence comparisons and biological characteristics. J Gen Virol 1995; 76 (Pt 7): 1571. 10. Daniel AM, Swenson JJ, Mayreddy RP, Khalili K, Frisque RJ. Sequences within the early and late promoters of archetype JC virus restrict viral DNA replication and infectivity. Virology 1996; 216 (1): 90. 11. Randhawa PS, Zygmunt D, Weck K, Shapiro R, Vats A, Swalsky P, Finkelstein S. Viral regulatory region seq rearrangements in renal allograft tissue from patients with BK virus nephropathy. Kid Internat 2003; 64: 743-47. 12. Stoner GL, Alappan R, Jobes DV, Ryschkewitsch CF, Landry ML. BK virus regulatory region rearrangements in brain and cerebrospinal fluid from a leukemia patient with tubulointerstitial nephritis and meningoencephalitis. Am J Kidney Dis 2002; 39 (5): 1102. 13. Carbone M, Rizzo P, Grimley PM, et al. Simian virus-40 large-T antigen binds p53 in human mesotheliomas. Nat Med 1997; 3 (8): 908. 14. De Luca A, Baldi A, Esposito V, et al. The retinoblastoma gene family pRb/p105, p107, pRb2/p130 and simian virus-40 large T-antigen in human mesotheliomas. Nat Med 1997; 3 (8): 913. 15. Hahn WC, Dessain SK, Brooks MW, et al. Enumeration of the simian virus 40 early region elements necessary for human cell transformation. Mol Cell Biol 2002; 22 (7): 2111. 16. Geetha D, Tong BC, Racusen L, Markowitz JS, Westra WH. Bladder carcinoma in a transplant recipient: evidence to implicate the BK human polyomavirus as a causal transforming agent. Transplantation 2002; 73 (12): 1933. 17. Monini P, Rotola A, Di Luca D, et al. DNA rearrangements impairing BK virus productive infection in urinary tract tumors. Virology 1995; 214 (1): 273. 18. Knepper JE, diMayorca G. Cloning and characterization of BK virus-related DNA sequences from normal and neoplastic human tissues. J Med Virol 1987; 21 (3): 289. 19. Arrington AS, Lednicky JA, Butel JS. Molecular characterization of SV40 DNA in multiple samples from a human mesothelioma [see comments]. Anticancer Res 2000; 20 (2A): 879. 20. Butel JS. Increasing evidence for involvement of SV40 in human cancer. Dis Markers 2001; 17 (3): 167. 21. Yanagihara R, Nerurkar VR, Scheirich I, et al. JC virus genotypes in the western Pacific suggest Asian mainland relationships and virus association with early population movements. Hum Biol 2002; 74 (3): 473. 22. Jin L. Rapid genomic typing of BK virus directly from clinical specimens. Molecular & Cellular Probes 1993; 7 (4): 331. 23. Jin L, Gibson PE. Genomic Function and Variation of Human Polyomavirus BK (BKV). Rev Med Virol 1996; 6 (4): 201. 24. Jin L, Gibson PE, Booth JC, Clewley JP. Genomic typing of BK virus in clinical specimens by direct sequencing of polymerase chain reaction products. Journal of Medical Virology 1993; 41 (1): 11. 25. Knowles WA. The epidemiology of BK virus and the occurrence of antigenic and genomic subtypes. In: Stoner GL, ed. Human polyomaviruses: molecular and clinical perspectives. New York: Wiley-Liss, 2001: 527. 26. Knowles WA, Gibson PE, Gardner SD. Serological typing scheme for BK-like isolates of human polyomavirus. J Med Virol 1989; 28 (2): 118. 27. Agostini HT, Ryschkewitsch CF, Baumhefner RW, et al. Influence of JC virus coding region genotype on risk of multiple sclerosis and progressive multifocal leukoencephalopathy. J Neurovirol 2000; 6 Suppl 2: S101. 28. Agostini HT, Ryschkewitsch CF, Mory R, Singer EJ, Stoner GL. JC virus (JCV) genotypes in brain tissue from patients with progressive multifocal leukoencephalopathy (PML) and in urine from controls without PML: increased frequency of JCV type 2 in PML. J Infect Dis 1997; 176 (1): 1. 29. Agostini HT, Ryschkewitsch CF, Stoner GL. Genotype profile of human polyomavirus JC excreted in urine of immunocompetent individuals. J Clin Microbiol 1996; 34 (1): 159. 30. Agostini HT, Ryschkewitsch CF, Stoner GL. Complete genome of a JC virus genotype type 6 from the brain of an African American with progressive multifocal leukoencephalopathy. J Hum Virol 1998; 1 (4): 267. 31. Agostini HT, Shishidohara Y, Baumhefner RW, Singer EJ, Ryschkewitsch CF, Stoner GL. Jc Virus Type 2 - Definition of Subtypes Based On Dna Sequence Analysis of Ten Complete Genomes. Journal of General Virology 1998; 79 (Part 5): 1143. 32. Agostini HT, Yanagihara R, Davis V, Ryschkewitsch CF, Stoner GL. Asian Genotypes of JC Virus in Native Americans and in a Pacific Island Population - Markers of Viral Evolution and Human Migration. Proceedings of the National Academy of Sciences of the United States of America 1997; 94 (26): 14542. 33. Jobe DV, Friedlaender JS, Mgone CS, et al. New JC virus (JCV) genotypes from papua new guinea and micronesia (type 8 and type 2E) and evolutionary analysis of 32 complete JCV genomes. Arch Virol 2001; 146 (11): 2097. 34. Bauer PH, Bronson RT, Fung SC, et al. Genetic and structural analysis of a virulence determinant in polyomavirus VP1. J Virol 1995; 69 (12): 7925. 35. Mannova P, Liebl D, Krauzewicz N, et al. Analysis of mouse polyomavirus mutants with lesions in the minor capsid proteins. J Gen Virol 2002; 83 (Pt 9): 2309. 36. Resnick J, Shenk T. Simian virus 40 agnoprotein facilitates normal nuclear location of the major capsid polypeptide and cell-to-cell spread of virus. J Virol 1986; 60 (3): 1098. 37. Carswell S, Alwine JC. Simian virus 40 agnoprotein facilitates perinuclear-nuclear localization of VP1, the major capsid protein. J Virol 1986; 60 (3): 1055. 38. Ng SC, Mertz JE, Sanden-Will S, Bina M. Simian virus 40 maturation in cells harboring mutants deleted in the agnogene. J Biol Chem 1985; 260 (2): 1127. 39. Del Valle L, Azizi SA, Krynska B, Enam S, Croul SE, Khalili K. Reactivation of human neurotropic JC virus expressing oncogenic protein in a recurrent glioblastoma multiforme. Ann Neurol 2000; 48 (6): 932. 40. Shah K, Nathanson N. Human exposure to SV40: review and comment. Am J Epidemiol 1976; 103 (1): 1. 41. Butel JS, Jafar S, Stewart AR, Lednicky JA. Detection of authentic SV40 DNA sequences in human brain and bone tumours. Dev Biol Stand 1998; 94: 23. 42. Kim JY, Koralnik IJ, LeFave M, Segal RA, Pfister LA, Pomeroy SL. Medulloblastomas and primitive neuroectodermal tumors rarely contain polyomavirus DNA sequences. Neuro-oncol 2002; 4 (3): 165. 43. Klein G, Powers A, Croce C. Association of SV40 with human tumors. Oncogene 2002; 21 (8): 1141. 44. Lednicky JA, Garcea RL, Bergsagel DJ, Butel JS. Natural simian virus 40 strains are present in human choroid plexus and ependymoma tumors. Virology 1995; 212 (2): 710. 45. Malkin D. Simian virus 40 and non-Hodgkin lymphoma. Lancet 2002; 359 (9309): 812. 46. Shivapurkar N, Harada K, Reddy J, et al. Presence of simian virus 40 DNA sequences in human lymphomas. Lancet 2002; 359 (9309): 851. 47. Li RM, Branton MH, Tanawattanacharoen S, Falk RA, Jennette JC, Kopp JB. Molecular Identification of SV40 Infection in Human Subjects and Possible Association with Kidney Disease. J Am Soc Nephrol 2002; 13 (9): 2320. 48. Butel JS, Arrington AS, Wong C, Lednicky JA, Finegold MJ. Molecular evidence of simian virus 40 infections in children. J Infect Dis 1999; 180 (3): 884. 49. Sundsfjord A, Osei A, Rosenqvist H, et al. BK and JC viruses in patients with systemic lupus erythematosus: prevalent and persistent BK viruria, sequence stability of the viral regulatory regions, and nondetectable viremia. J Infect Dis 1999; 180 (1): 1. 50. Dubois V, Dutronc H, Lafon ME, et al. Latency and reactivation of JC virus in peripheral blood of human immunodeficiency virus type 1-infected patients. J Clin Microbiol 1997; 35 (9): 2288. 51. Ferrante P, Caldarelli-Stefano R, Omodeo-Zorini E, et al. Comprehensive investigation of the presence of JC virus in AIDS patients with and without progressive multifocal leukoencephalopathy. J Med Virol 1997; 52 (3): 235. 52. Perrons CJ, Fox JD, Lucas SB, Brink NS, Tedder RS, Miller RF. Detection of polyomaviral DNA in clinical samples from immunocompromised patients: correlation with clinical disease. J Infect 1996; 32 (3): 205. 53. Major EO, Curfman BL. Viral induced demyelination leading to progressive multifocal leukoencephalopathy: the involvement of both immune and nervous system target cells. In: Remington JS, ed. In Defense of the Brain. Boston, MA: Blackwell Science, 1997: 305. 54. Gallia GL, Houff SA, Major EO, Khalili K. Review: JC virus infection of lymphocytes--revisited. J Infect Dis 1997; 176 (6): 1603. 55. Monaco MC, Atwood WJ, Gravell M, Tornatore CS, Major EO. JC virus infection of hematopoietic progenitor cells, primary B lymphocytes, and tonsillar stromal cells: implications for viral latency. J Virol 1996; 70 (10): 7004. 56. Lafon ME, Dutronc H, Dubois V, et al. Jc Virus Remains Latent in Peripheral Blood B Lymphocytes But Replicates Actively in Urine From Aids Patients. Journal of Infectious Diseases 1998; 177 (6): 1502. 57. Kitamura T, Sugimoto C, Kato A, et al. Persistent JC virus (JCV) infection is demonstrated by continuous shedding of the same JCV strains. J Clin Microbiol 1997; 35 (5): 1255. 58. Chang D, Wang M, Ou WC, Lee MS, Ho HN, Tsai RT. Genotypes of human polyomaviruses in urine samples of pregnant women in Taiwan. J Med Virol 1996; 48 (1): 95. 59. Stoner GL, Agostini HT, Ryschkewitsch CF, Baumhefner RW, Tourtellotte WW. Characterization of JC virus DNA amplified from urine of chronic progressive multiple sclerosis patients. Mult Scler 1996; 1 (4): 193. 60. Markowitz RB, Thompson HC, Mueller JF, Cohen JA, Dynan WS. Incidence of BK virus and JC virus viruria in human immunodeficiency virus-infected and -uninfected subjects. Journal of Infectious Diseases 1993; 167 (1): 13. 61. Koralnik IJ, Boden D, Mai VX, Lord CI, Letvin NL. JC virus DNA load in patients with and without progressive multifocal leukoencephalopathy. Neurology 1999; 52 (2): 253. 62. Worthmann F, Turker T, Muller AR, Patt S, Stoltenburg-Didinger G. Progressive multifocal leukoencephalopathy after orthotopic liver transplantation. Transplantation 1994; 57 (8): 1268. 63. Hair LS, Nuovo G, Powers JM, Sisti MB, Britton CB, Miller JR. Progressive multifocal leukoencephalopathy in patients with human immunodeficiency virus. Hum Pathol 1992; 23 (6): 663. 64. Jeong BH, Jin JK, Choi EK, et al. Detection of JC Virus Type 1 in Peripheral Lymphocytes, Brain and Cerebrospinal Fluid from Two Korean AIDS Patients with Progressive Multifocal Leukoencephalopathy. Intervirology 2002; 45 (2): 94. 65. Randhawa P, Baksh F, Aoki N, Tschirhart D, Finkelstein S. JC virus infection in allograft kidneys: analysis by polymerase chain reaction and immunohistochemistry. Transplantation 2001; 71 (9): 1300. 66. Gardner SD, Field AM, Coleman DV, Hulme B. New human papovavirus (B.K.) isolated from urine after renal transplantation. Lancet 1971; 1 (7712): 1253. 67. Hogan TF, Borden EC, McBain JA, Padgett BL, Walker DL. Human polyomavirus infections with JC virus and BK virus in renal transplant patients. Annals of Internal Medicine 1980; 92 (3): 373. 68. Andrews CA, Shah KV, Daniel RW, Hirsch MS, Rubin RH. A serological investigation of BK virus and JC virus infections in recipients of renal allografts. Journal of Infectious Diseases 1988; 158 (1): 176. 69. Gardner SD, MacKenzie EF, Smith C, Porter AA. Prospective study of the human polyomaviruses BK and JC and cytomegalovirus in renal transplant recipients. Journal of Clinical Pathology 1984; 37 (5): 578. 70. Rosen S, Harmon W, Krensky AM, et al. Tubulo-interstitial nephritis associated with polyomavirus (BK type) infection. N Engl J Med 1983; 308 (20): 1192. 71. Vallbracht A, Lohler J, Gossmann J, et al. Disseminated BK type polyomavirus infection in an AIDS patient associated with central nervous system disease. Am J Pathol 1993; 143 (1): 29. 72. Randhawa PS, Finkelstein S, Scantlebury V, et al. Human polyoma virus-associated interstitial nephritis in the allograft kidney. Transplantation 1999; 67 (1): 103. 73. Randhawa PS, Demetris AJ. Nephropathy due to polyomavirus type BK. N Engl J Med 2000; 342 (18): 1361. 74. Hirsch HH, Knowles W, Dickenmann M, et al. Prospective study of polyomavirus type BK replication and nephropathy in renal-transplant recipients. N Engl J Med 2002; 347 (7): 488. 75. Nickeleit V, Hirsch HH, Binet IF, et al. Polyomavirus infection of renal allograft recipients: from latent infection to manifest disease. J Am Soc Nephrol 1999; 10 (5): 1080. 76. Drachenberg CB, Beskow CO, Cangro CB, et al. Human polyoma virus in renal allograft biopsies: Morphological findings and correlation with urine cytology. Human Pathology 1999; 30 (8): 970. 77. Mylonakis E, Goes N, Rubin RH, Cosimi AB, Colvin RB, Fishman JA. Bk virus in solid organ transplant recipients: an emerging syndrome. Transplantation 2001; 72 (10): 1587. 78. Drachenberg RC, Drachenberg CB, Papadimitriou JC, et al. Morphological spectrum of polyoma virus disease in renal allografts: diagnostic accuracy of urine cytology. Am J Transplant 2001; 1 (4): 373. 79. Ramos E, Drachenberg CB, Papadimitriou JC, et al. Clinical course of polyoma virus nephropathy in 67 renal transplant patients. J Am Soc Nephrol 2002; 13 (8): 2145. 80. Humar A, Gillingham KJ, Payne WD, Dunn DL, Sutherland DE, Matas AJ. Association between cytomegalovirus disease and chronic rejection in kidney transplant recipients. Transplantation 1999; 68 (12): 1879. 81. Lewis RM, Johnson PC, Golden D, Van Buren CT, Kerman RH, Kahan BD. The adverse impact of cytomegalovirus infection on clinical outcome in cyclosporine-prednisone treated renal allograft recipients. Transplantation 1988; 45 (2): 353. 82. Atencio IA, Shadan FF, Zhou XJ, Vaziri ND, Villarreal LP. Adult mouse kidneys become permissive to acute polyomavirus infection and reactivate persistent infections in response to cellular damage and regeneration. Journal of Virology 1993; 67 (3): 1424. 83. Nickeleit V, Klimkait T, Binet IF, et al. Testing for polyomavirus type BK DNA in plasma to identify renal-allograft recipients with viral nephropathy. N Engl J Med 2000; 342 (18): 1309. 84. Hirsch HH, et al. Prospective study of polyomavirus type BK replication and nephropathy in renal transplant recipients. N Engl J Med 2002; 347: 488. 85. Randhawa P, Ho A, Vats A, Shapiro R, Weck K. Quantitative measurement of BK virus load in urine and plasma of renal transplant patients: correlation with biopsy findings. Am J Transplant 2003; 3 (supplement 5): 371A. 86. Vats A, Shapiro R, Randhawa P et al. BK virus associated nephropathy and cidofovir: long term experience. Am J Transplant 2003; 3 (supplement 5): 190A. 87. Russell JK, Blalock JE. Vitamin A inhibition of polyoma virus replication. Biochem Biophys Res Commun 1984; 122 (2): 851. 88. Ferrazzi E, Peracchi M, Biasolo MA, Faggionato O, Stefanelli S, Palu G. Antiviral activity of gyrase inhibitors norfloxacin, coumermycin A1 and nalidixic acid. Biochem Pharmacol 1988; 37 (9): 1885. 89. Kerr DA, Chang CF, Gordon J, Bjornsti MA, Khalili K. Inhibition of human neurotropic virus (JCV) DNA replication in glial cells by camptothecin. Virology 1993; 196 (2): 612. 90. Poduval RD, Kadambi P, Javaid B et al. Leflunomide – a potential new therapeutic agent for BKV nephropathy. Am J Transplant 2003; 3 (supplement 5): 189A |