
|
Paneles de Discussión
Paneais de Discussio |
Hypertension and Salt-sensitivity. Pathophysiology and Clinical Implications.Alejandro de la Sierra MD. PhD., Ernesto Bragulat MD., María Larrousse MD.Hypertension Unit. Department of Internal Medicine.
|
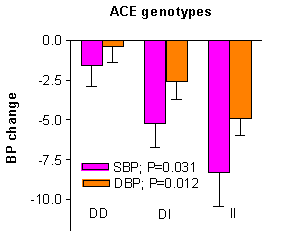 |  |
Neurohumoral responses to salt intake or depletion also vary between salt sensitive and salt resistant hypertensives, with a blunted response in RAS activity and a paradoxical increase in neurotransmitters in response to salt intake being reported in the former (figure 2)10. Intracellular sodium metabolism abnormalities also occur in salt sensitive patients, especially an increased erythrocyte Na/Li countertransport activity11.

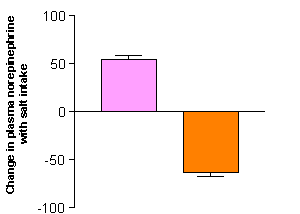
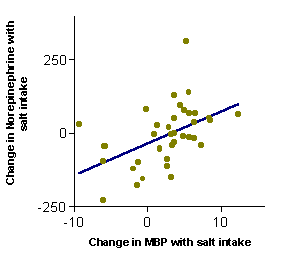
More recently, it has been suggested that functional vascular abnormalities involving increased levels of early markers of atherosclerosis are more pronounced in salt sensitive hypertensive subjects. Over the last few years, our group has studied functional vascular abnormalities in salt sensitive hypertension. We have investigated several parameters of vascular function, including endothelial dysfunction, oxidative stress and inflammation markers, which could represent the expression of an accelerated atherosclerotic process and thus explain the increased number of cardiovascular complications reported in salt sensitive subjects.
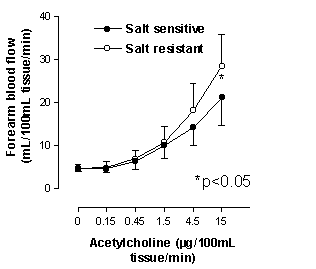
We have studied a group of essential hypertensive patients, classified on the basis of their salt sensitivity or resistance. Endothelium-derived factors, such as nitrates and nitrites – as indirect measures of nitric oxide (NO) production, cyclic GMP - as the intermediate mediator of NO actions, and endothelin, were measured at both low and high salt intakes, as were the variations induced by a high salt diet. The results showed that whereas high salt intake tended to reduce NO production (a reduction in both plasma and urinary nitrates and nitrites concentration), this was more clearly evident in the salt sensitive subset. Urinary nitrate excretion was reduced by high salt intake in salt sensitive patients, whereas NO production was increased in salt resistant subjects. These subtle alterations in endothelial function observed in salt sensitive subjects were more clearly demonstrated by studying the endothelium-dependent vascular responses "in vivo" in the forearm arteries. The infusion of increasing doses of both acetylcholine, a substance which mediates an endothelium-dependent vasodilation, and sodium nitroprusside, which mediates an endothelium-independent vasodilation, resulted in salt sensitive hypertensive subjects presenting significantly lower maximal acetylcholine-induced vasodilation compared with salt resistant hypertensive patients (figure 3). Conversely, maximal sodium nitroprusside-induced vasodilation did not differ between groups. This endothelium-dependent impaired response in salt sensitive subjects was accompanied by a lack of effect of L-NMMA administration, a specific NO synthase inhibitor, which reduced maximal acetylcholine-induced vasodilation in salt resistant but not in salt sensitive subjects12.
 |  |
Alterations in endothelial function reported in salt sensitive hypertensive subjects emphasize the importance of NO in regulating vascular tone. Abnormalities in endothelial function have been reported in hypertension, diabetes and dyslipidemia, and are constant features in the early stages of the atherosclerotic process13. Moreover, greater endothelial dysfunction may be related to metabolic abnormalities, which are also present in salt sensitive subjects. Indeed, low HDL-cholesterol, high LDL-cholesterol, high plasma fasting insulin, glucose intolerance and insulin resistance are all present or more pronounced in salt sensitive patients14,15.
Other important features related to the development of the atherosclerotic process are excessive oxidative stress and increased serum and tissue markers of inflammation, cell adhesion, migration and proliferation, and matrix deposition and degradation. An interesting hypothesis is that salt sensitivity may be related to an increase in oxidative stress and/or inflammation markers. Although preliminary, results in a small group of hypertensive subjects support the idea that both processes are enhanced in salt sensitivity. In fact, salt sensitive hypertensives tend to present higher levels of homocysteine and reduced levels of serum antioxidants such as selenium and vitamin C, suggesting an increase in oxidative stress16. It is known that the production of reactive oxygen species counteracts the vasodilating, antiproliferative and antiagregant actions of NO by rapidly degrading it to peroxynitrites. The reduction in these antioxidant agents may be related to a low NO bioavailability and thus to the endothelial dysfunction reported in salt sensitive individuals.
We have recently found increased serum levels of several markers of vascular inflammation to be related to salt sensitivity. Compared to salt resistant hypertensives, salt sensitive subjects present higher levels of serum intracellular adhesion molecule-1 (ICAM-1), P-selectin and macrophage attractant chemoprotein-1 (MCP-1), which represent an enhanced inflammatory response in the vascular wall. Salt sensitive hypertensives also show lower serum levels of matrix metalloproteinase type 9 (MMP-9) and the tissue inhibitor of metalloproteinases type 1 (TIMP-1)17. MMP-9 is one of the metalloproteinases involved in the digestion of the extracellular matrix that accompanies vascular hypertrophy. TIMP-1 inhibits the digestion of this extracellular matrix, and a close association with left ventricular hypertrophy in essential hypertensive patients has been recently reported18. A lack of equilibrium in plasma factors involved in the extracellular matrix formation and degradation observed in salt sensitive subjects could represent a link between salt intake, salt sensitivity, hypertension, and vascular and cardiac hypertrophy, thus leading to a higher incidence of hypertension-related cardiovascular complications in salt sensitive subjects.
In this sense, salt sensitive hypertensive subjects present an increased incedence of target organ damage, including microalbuminuria19 and left ventricular hypertrophy14. Moreover, two independent groups have found increased rates of cardiovascular complication in salt sensitive subjects, both hypertensives and normotensives20,21.
References